Organic chemistry might seem daunting, but it's actually like learning... Show more
A Level Chemistry Module 4 OCR Organic Revision Notes









Module 4 Overview
This is your guide to mastering organic chemistry fundamentals. You'll learn about the two main types of hydrocarbons and their key reactions.
The content focuses on alkanes (saturated hydrocarbons) and alkenes (unsaturated hydrocarbons), which form the foundation for understanding more complex organic molecules.

Key Terms and Definitions
Understanding organic chemistry starts with mastering the vocabulary - these terms will come up repeatedly in your exams. Hydrocarbons are simply compounds made of only hydrogen and carbon atoms, whilst functional groups are the reactive parts that give molecules their characteristic properties.
Saturated compounds have only single carbon-carbon bonds, making them quite stable. In contrast, unsaturated compounds contain double or triple bonds, making them much more reactive.
A homologous series is like a family of compounds - each member has the same functional group but differs by CH₂. Think of it as a pattern that makes organic chemistry predictable rather than random.
Quick Tip: Remember that aromatic compounds contain benzene rings, whilst aliphatic compounds are everything else - it's that simple!

Alkanes - The Stable Hydrocarbons
Alkanes are the "boring" molecules of organic chemistry, but that's exactly what makes them useful as fuels. They're saturated hydrocarbons with only sigma bonds, which allow free rotation around each carbon atom.
The tetrahedral shape around each carbon (109.5°) comes from four bonding pairs of electrons repelling equally. As alkane chains get longer, their boiling points increase because stronger induced dipole-dipole interactions require more energy to overcome.
Branched alkanes have lower boiling points than straight-chain ones because branching reduces surface contact between molecules, weakening intermolecular forces. This explains why petrol (highly branched) evaporates more easily than diesel.
Alkanes undergo complete combustion with plenty of oxygen to produce CO₂ and H₂O, but incomplete combustion in limited oxygen produces dangerous carbon monoxide and soot particles.
Exam Alert: You'll often be asked to explain boiling point trends - remember it's all about intermolecular forces and surface contact!

Radical Substitution with Halogens
When alkanes meet halogens under UV light, things get interesting through radical substitution. This three-stage mechanism involves highly reactive radicals - atoms or molecules with unpaired electrons.
Initiation starts when UV light breaks the halogen molecule by homolytic fission, creating two radical atoms. Propagation involves chain reactions where radicals react to form products whilst generating new radicals to continue the process.
Termination occurs when two radicals combine to form stable molecules, ending the chain reaction. The problem with this reaction is that substitution can happen anywhere on the carbon chain, creating multiple products.
For your exams, you need to write mechanisms clearly. Start with initiation (showing homolytic fission with UV), then show two propagation steps, and finish with possible termination reactions.
Memory Trick: Think of radical substitution as a chain reaction - once it starts, it keeps going until radicals run out!

Alkenes - The Reactive Double Bond
Alkenes are far more exciting than alkanes because of their pi bond, which sits above and below the main carbon-carbon bond. These pi electrons are easily accessible, making alkenes perfect for addition reactions.
Hydrogenation converts alkenes to alkanes using hydrogen gas, a nickel catalyst, and 150°C. Halogenation happens at room temperature and provides a useful test - bromine water decolourises when it reacts with alkenes.
Hydrogen halides add across the double bond at room temperature to form haloalkanes. Hydration uses steam with concentrated acid catalysts above 100°C to make alcohols.
All these reactions follow the same pattern - the pi bond breaks and two atoms or groups add across the original C=C. This predictability makes alkenes incredibly useful in industrial chemistry.
Lab Test: The bromine water test is your go-to method for detecting alkenes - orange bromine turns colourless!
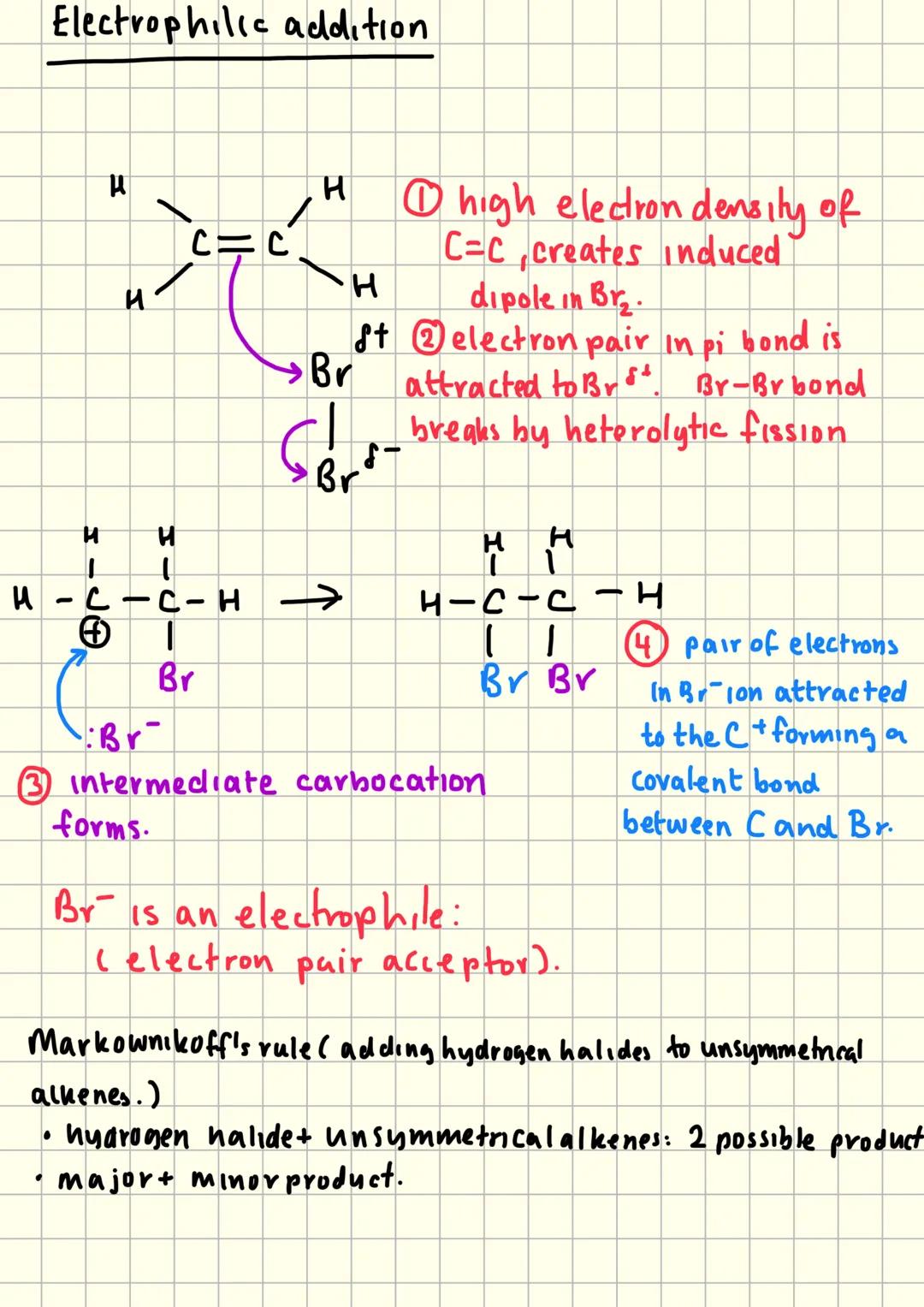
Electrophilic Addition Mechanism
Electrophilic addition explains how alkenes react with polar molecules like hydrogen halides. The process starts when the electron-rich C=C bond attracts electron-deficient species called electrophiles.
Step one involves the pi electrons attacking the electrophile, causing heterolytic fission of the attacking molecule. This creates a carbocation intermediate and a negative ion.
Step two sees the negative ion attacking the positive carbocation to form the final product. The whole process happens because alkenes have high electron density that can induce dipoles in approaching molecules.
Markovnikov's rule becomes crucial with unsymmetrical alkenes - the hydrogen atom adds to the carbon with the most hydrogens already attached, giving major and minor products.
Exam Focus: Draw mechanisms clearly with curly arrows showing electron movement - this is where marks are easily lost or gained!
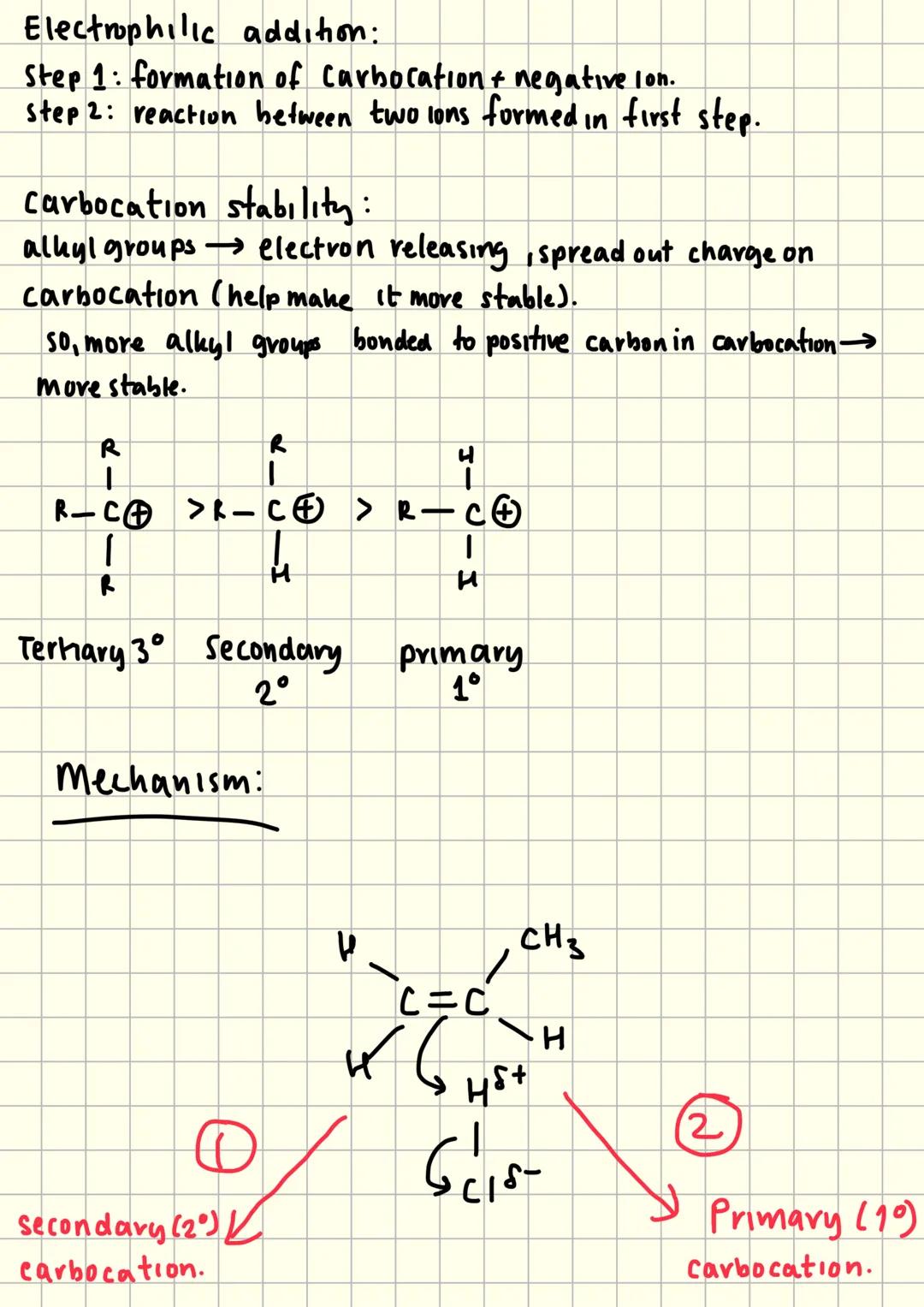
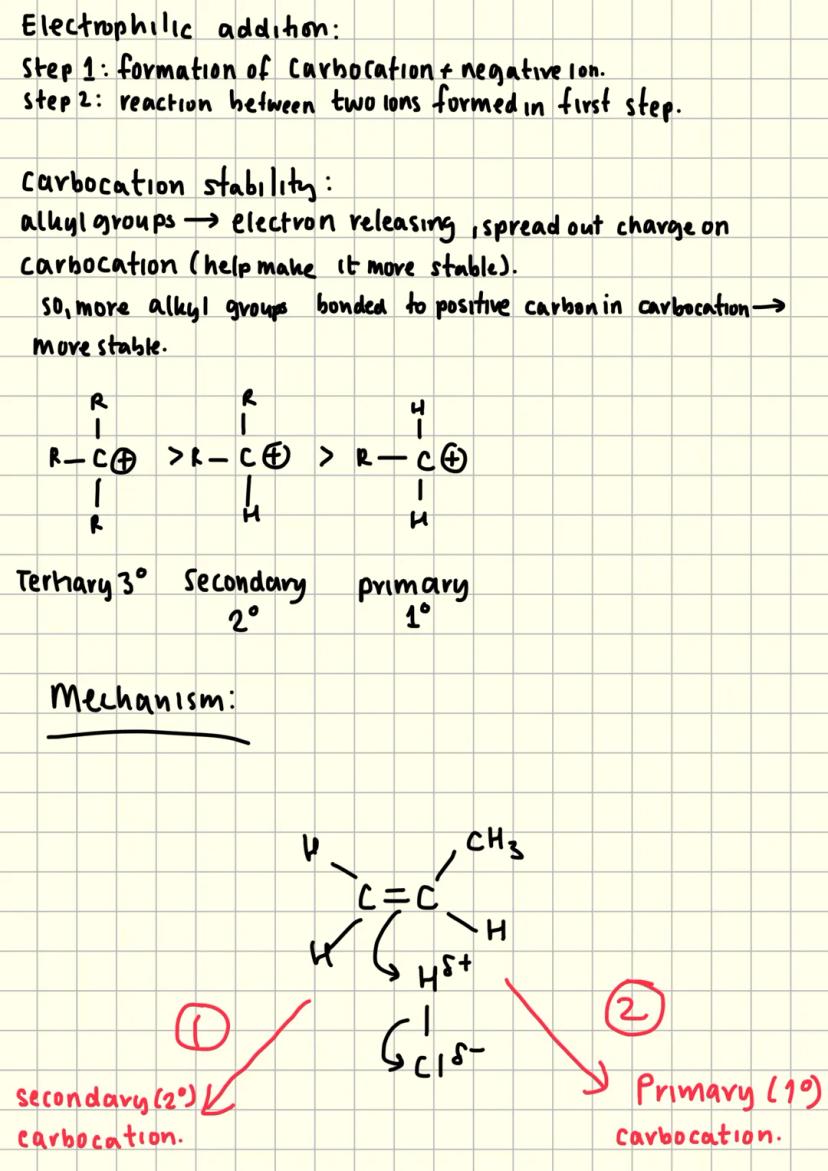
Carbocation Stability and Markovnikov's Rule
Carbocation stability determines which products form during electrophilic addition. Alkyl groups are electron-releasing, which spreads out the positive charge and stabilises the carbocation.
Tertiary carbocations (3°) are most stable because they have three alkyl groups spreading the charge. Secondary (2°) are less stable, whilst primary (1°) carbocations are least stable.
The major product always forms via the more stable carbocation intermediate. This explains Markovnikov's rule - addition occurs to give the most stable carbocation possible.
Understanding this concept helps predict products in exam questions. Look for which carbocation would be more stable, and you'll know which product predominates.
Quick Check: Count the alkyl groups attached to the positive carbon - more groups mean more stability!

Markovnikov's Rule in Action
This page shows Markovnikov's rule working with a specific example. When HCl adds to an unsymmetrical alkene, two different carbocations can form, but one is much more stable.
The secondary carbocation forms the major product because it's more stable than the primary carbocation. This isn't a 50:50 split - the major product dominates significantly.
In exam questions, you'll need to identify which carbocation is more stable and predict the major product accordingly. Remember that the hydrogen always adds to create the more stable carbocation.
Exam Strategy: When predicting products, always consider both possible carbocations and choose the more stable pathway!
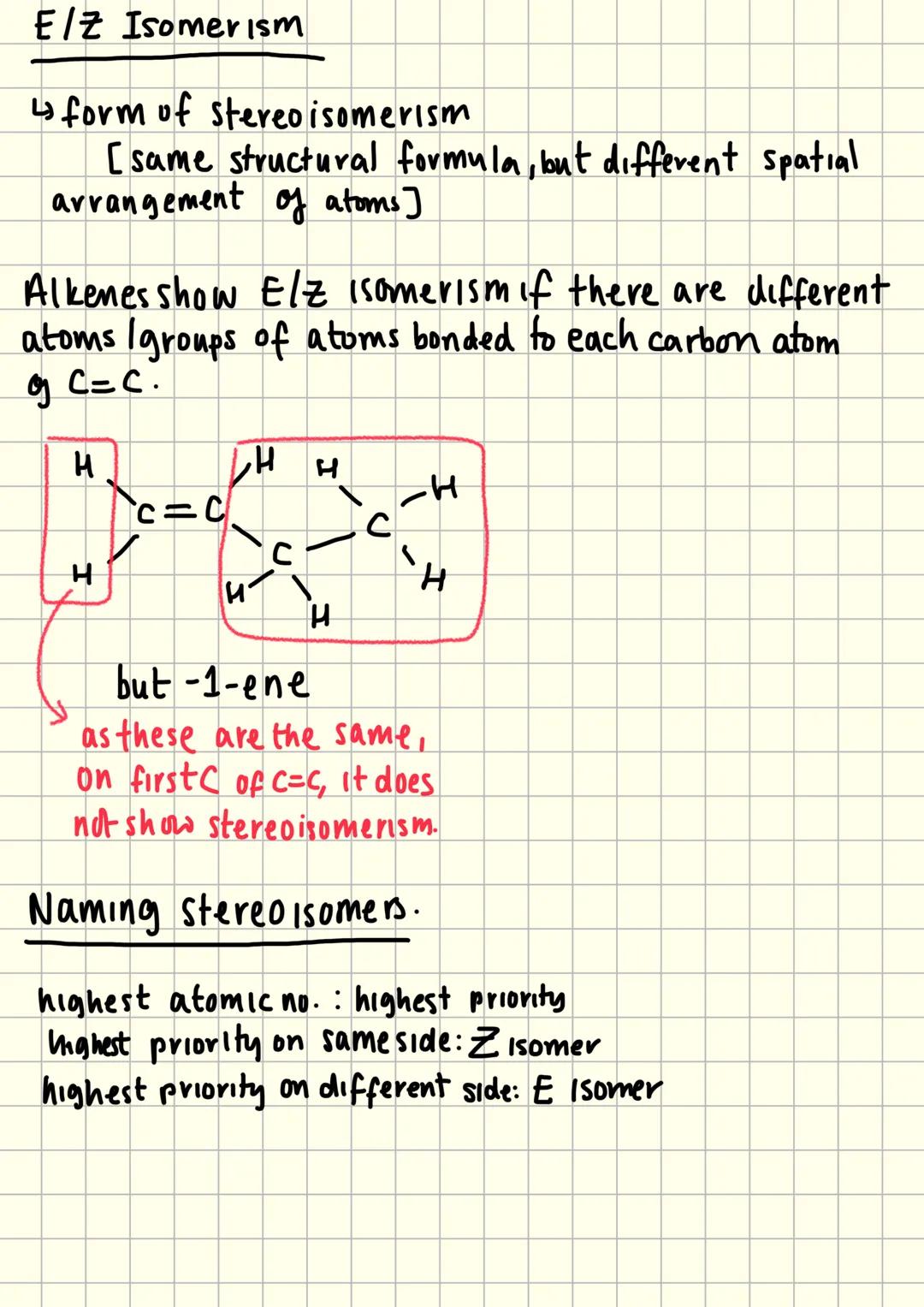
E/Z Isomerism Basics
E/Z isomerism is a type of stereoisomerism where molecules have the same structural formula but different spatial arrangements. This only occurs when different atoms or groups attach to each carbon of a C=C double bond.
The restricted rotation around double bonds means these different arrangements are actually separate compounds with different properties. Both carbons of the C=C must have different groups attached - if any carbon has two identical groups, no stereoisomerism exists.
Naming stereoisomers uses priority rules based on atomic number. Higher atomic numbers get higher priority, and the arrangement of high-priority groups determines the name.
Z isomers have the highest priority groups on the same side of the double bond, whilst E isomers have them on opposite sides.
Memory Aid: Think "Z = Zame zide" and "E = opposite Ends" to remember the naming system!
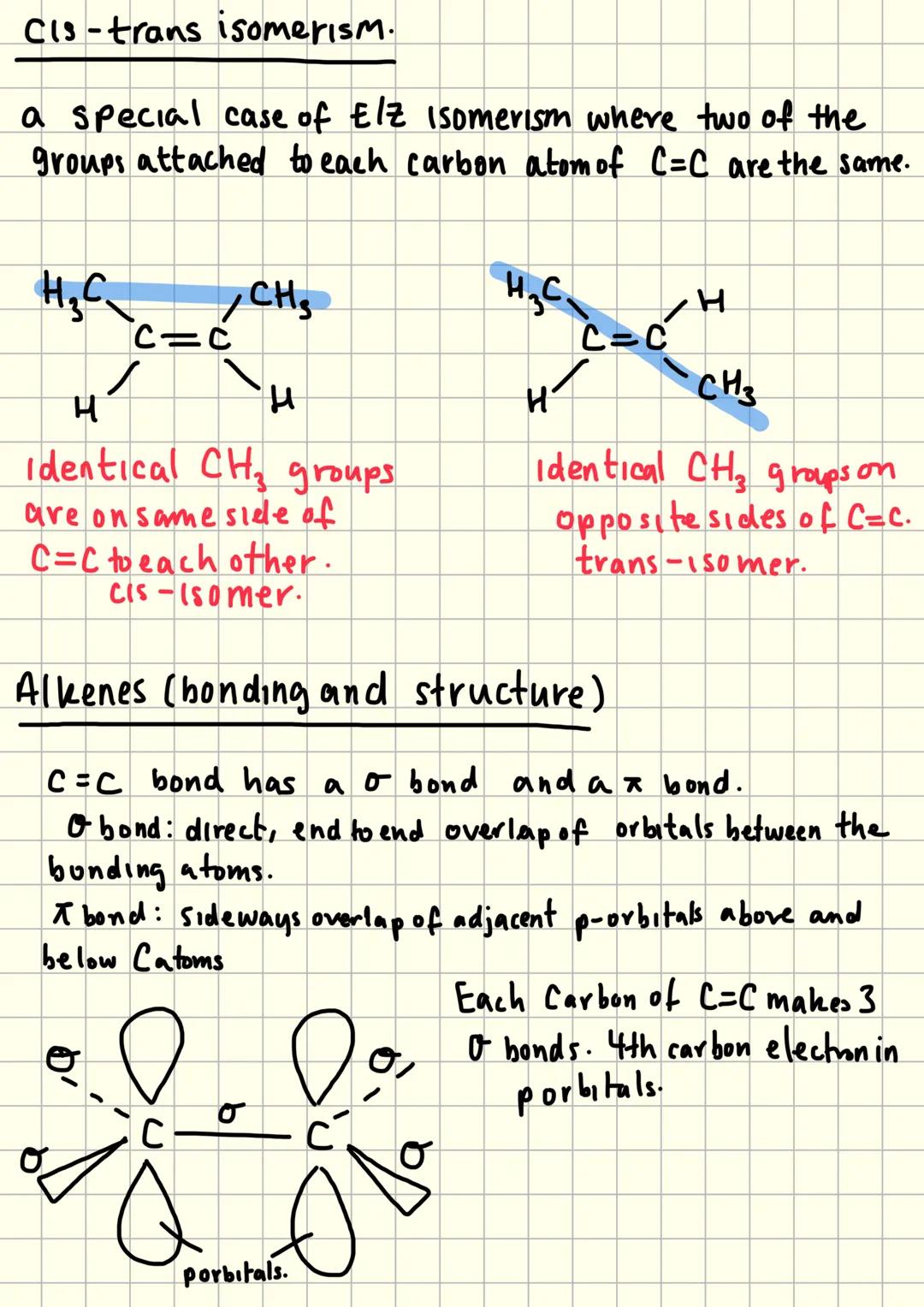
Cis-Trans Isomerism and Bonding
Cis-trans isomerism is a special case of E/Z isomerism where two identical groups attach to each carbon of the C=C. Cis means identical groups are on the same side, whilst trans means they're on opposite sides.
The C=C double bond contains both a sigma bond and a pi bond. Sigma bonds form from direct end-to-end orbital overlap, whilst pi bonds form from sideways overlap of p-orbitals above and below the bonding atoms.
Each carbon in C=C makes three sigma bonds using sp² hybridisation. The fourth electron occupies a p-orbital, and these p-orbitals overlap sideways to form the pi bond.
This bonding arrangement prevents rotation around the double bond, which is why stereoisomerism exists in alkenes but not alkanes.
Key Concept: The pi bond's sideways overlap prevents rotation - this restriction is what makes stereoisomerism possible!
We thought you’d never ask...
What is the Knowunity AI companion?
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
Where can I download the Knowunity app?
You can download the app from Google Play Store and Apple App Store.
Is Knowunity really free of charge?
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
Similar content
Most popular content: Organic Chemistry
9A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
Comprehensive Chemistry Overview
Master key chemistry concepts with this detailed cheat sheet covering topics like reaction rates, redox reactions, chromatography, and the periodic table. Perfect for exam preparation, this resource includes essential information on enthalpy changes, organic synthesis, and more. Ideal for A-Level students seeking to enhance their understanding and performance in chemistry.
Organic Chemistry Essentials
Explore key concepts in organic chemistry, including functional groups, IUPAC nomenclature, reaction mechanisms, and polymer chemistry. This comprehensive summary covers aromatic compounds, alkenes, aldehydes, ketones, and mass spectrometry, providing essential insights for students. Ideal for exam preparation and understanding organic synthesis, chemical reactions, and molecular structures.
chemistry spec topics checklist
ocr a level chemistry checklist made from the official specification
OCR A-Level Chemistry Vocabulary
Explore an extensive collection of key terms and concepts for OCR A-Level Chemistry, organized by topic and sub-topic. This resource is designed to enhance your understanding and application of advanced vocabulary in exams, covering essential areas such as atomic structure, organic chemistry, and reaction mechanisms.
Nat 5 chemistry unit 2
All necessary information for national 5 chemistry
Organic chemistry GCSE AQA
GCSE triple chemistry notes
Organic Reaction Mechanisms
Explore key organic reaction mechanisms including electrophilic addition, free radical substitution, and nucleophilic substitution. This summary covers hydrocarbons, alkanes, alkenes, and their transformations, essential for AQA AS Level organic chemistry. Ideal for students seeking to understand complex organic processes and improve exam performance.
Alkanes and Hydrocarbons Overview
Explore the fundamentals of alkanes and hydrocarbons in this concise guide. Understand the molecular and structural formulas, naming conventions, and the significance of isomers. Ideal for Year 12 students preparing for A-Level Organic Chemistry (OCR B Salters). Key concepts include saturated hydrocarbons, IUPAC nomenclature, and the characteristics of alkanes.
Most popular content in Chemistry
9GCSE AQA Chemistry Topic 1- The Atom
The Atom
chem paper 1
higher
A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
GCSE AQA Chemistry Topic 1
Quiz covering all of topic 1
Essential Chemistry Practicals
Explore key AQA GCSE Chemistry practicals, including flame tests, titration, and gas identification. This resource covers essential techniques for analyzing ions, making salts, and understanding reaction kinetics. Perfect for students preparing for exams and practical assessments.
chem paper 1
Unlock the secrets of chemical reactions with this comprehensive flashcard set designed to help you conquer complex concepts and ace your Chemistry exams.
AQA GCSE Chemistry 4.2 Bonding, structure and the Properties of Matter
4.2 Metals and Alloys
AQA GCSE Chemistry 4.4 Chemical changes
4.4 Reactions of acids with bases or carbonates
Fundamentals of Atomic Structure
Practice identifying subatomic particles, calculating neutrons, and defining isotopes and atomic/mass numbers.
Most popular content
9Sociology of Education Overview
Explore comprehensive A-Level Sociology notes on the education system, covering key theories, policies, and sociological perspectives. This resource includes insights on marketisation, gender roles, cultural deprivation, and educational inequalities, providing a thorough understanding of how education shapes social stratification and individual achievement. Ideal for exam preparation and in-depth study.
Sociology of Families: Comprehensive Revision
Dive into an extensive overview of family dynamics, perspectives, and patterns in sociology. This resource covers key concepts such as family diversity, gender roles, marriage, and the impact of social policies on family structures. Perfect for A-Level Sociology students preparing for Paper 2.
Criminology: Crime & Punishment Overview
Comprehensive mindmaps covering key concepts in the Crime and Punishment topic for WJEC Criminology Unit 4. This resource includes detailed insights into the Criminal Justice System, crime prevention strategies, sentencing models, and the roles of various agencies. Ideal for A-Level revision, ensuring you grasp essential theories and legislative processes to excel in your exams.
An Inspector Calls: Character Insights
Explore in-depth analysis and key quotes for characters in J.B. Priestley's 'An Inspector Calls'. This resource covers Gerald Croft, Inspector Goole, Sheila Birling, Mrs. Birling, Eric Birling, and Eva Smith, focusing on themes of class, gender roles, and social responsibility. Ideal for students aiming for Grade 8 and above.
WJEC Unit 4 Criminology
Criminology unit 4 detailed revision note
Criminology Theories Overview
Explore key criminology theories and their implications on crime and deviance. This comprehensive summary covers biological, psychological, and sociological perspectives, including labelling theory, right realism, and the impact of social campaigns on policy development. Ideal for A-Level criminology students seeking to understand the complexities of criminal behaviour and the factors influencing crime prevention strategies.
Romeo and Juliet: Key themes
Key Romeo and Juliet themes and analysed quotes
Macbeth: Guilt and Ambition
Explore the complex themes of guilt and ambition in Shakespeare's 'Macbeth'. This analysis covers key characters, including Macbeth and Lady Macbeth, their moral dilemmas, and the tragic consequences of their ambition. Ideal for students studying character motivations, thematic elements, and the psychological impact of power. Includes insights on the natural order, manipulation, and the descent into madness.
Cell Biology and Cell structure
cell structures
Can't find what you're looking for? Explore other subjects.
Students love us — and so will you.
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
A Level Chemistry Module 4 OCR Organic Revision Notes
Organic chemistry might seem daunting, but it's actually like learning a language with predictable patterns and rules. This module covers the fundamental building blocks of organic compounds - alkanes and alkenes - and how they behave in different reactions.

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Module 4 Overview
This is your guide to mastering organic chemistry fundamentals. You'll learn about the two main types of hydrocarbons and their key reactions.
The content focuses on alkanes (saturated hydrocarbons) and alkenes (unsaturated hydrocarbons), which form the foundation for understanding more complex organic molecules.

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Key Terms and Definitions
Understanding organic chemistry starts with mastering the vocabulary - these terms will come up repeatedly in your exams. Hydrocarbons are simply compounds made of only hydrogen and carbon atoms, whilst functional groups are the reactive parts that give molecules their characteristic properties.
Saturated compounds have only single carbon-carbon bonds, making them quite stable. In contrast, unsaturated compounds contain double or triple bonds, making them much more reactive.
A homologous series is like a family of compounds - each member has the same functional group but differs by CH₂. Think of it as a pattern that makes organic chemistry predictable rather than random.
Quick Tip: Remember that aromatic compounds contain benzene rings, whilst aliphatic compounds are everything else - it's that simple!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Alkanes - The Stable Hydrocarbons
Alkanes are the "boring" molecules of organic chemistry, but that's exactly what makes them useful as fuels. They're saturated hydrocarbons with only sigma bonds, which allow free rotation around each carbon atom.
The tetrahedral shape around each carbon (109.5°) comes from four bonding pairs of electrons repelling equally. As alkane chains get longer, their boiling points increase because stronger induced dipole-dipole interactions require more energy to overcome.
Branched alkanes have lower boiling points than straight-chain ones because branching reduces surface contact between molecules, weakening intermolecular forces. This explains why petrol (highly branched) evaporates more easily than diesel.
Alkanes undergo complete combustion with plenty of oxygen to produce CO₂ and H₂O, but incomplete combustion in limited oxygen produces dangerous carbon monoxide and soot particles.
Exam Alert: You'll often be asked to explain boiling point trends - remember it's all about intermolecular forces and surface contact!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Radical Substitution with Halogens
When alkanes meet halogens under UV light, things get interesting through radical substitution. This three-stage mechanism involves highly reactive radicals - atoms or molecules with unpaired electrons.
Initiation starts when UV light breaks the halogen molecule by homolytic fission, creating two radical atoms. Propagation involves chain reactions where radicals react to form products whilst generating new radicals to continue the process.
Termination occurs when two radicals combine to form stable molecules, ending the chain reaction. The problem with this reaction is that substitution can happen anywhere on the carbon chain, creating multiple products.
For your exams, you need to write mechanisms clearly. Start with initiation (showing homolytic fission with UV), then show two propagation steps, and finish with possible termination reactions.
Memory Trick: Think of radical substitution as a chain reaction - once it starts, it keeps going until radicals run out!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Alkenes - The Reactive Double Bond
Alkenes are far more exciting than alkanes because of their pi bond, which sits above and below the main carbon-carbon bond. These pi electrons are easily accessible, making alkenes perfect for addition reactions.
Hydrogenation converts alkenes to alkanes using hydrogen gas, a nickel catalyst, and 150°C. Halogenation happens at room temperature and provides a useful test - bromine water decolourises when it reacts with alkenes.
Hydrogen halides add across the double bond at room temperature to form haloalkanes. Hydration uses steam with concentrated acid catalysts above 100°C to make alcohols.
All these reactions follow the same pattern - the pi bond breaks and two atoms or groups add across the original C=C. This predictability makes alkenes incredibly useful in industrial chemistry.
Lab Test: The bromine water test is your go-to method for detecting alkenes - orange bromine turns colourless!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Electrophilic Addition Mechanism
Electrophilic addition explains how alkenes react with polar molecules like hydrogen halides. The process starts when the electron-rich C=C bond attracts electron-deficient species called electrophiles.
Step one involves the pi electrons attacking the electrophile, causing heterolytic fission of the attacking molecule. This creates a carbocation intermediate and a negative ion.
Step two sees the negative ion attacking the positive carbocation to form the final product. The whole process happens because alkenes have high electron density that can induce dipoles in approaching molecules.
Markovnikov's rule becomes crucial with unsymmetrical alkenes - the hydrogen atom adds to the carbon with the most hydrogens already attached, giving major and minor products.
Exam Focus: Draw mechanisms clearly with curly arrows showing electron movement - this is where marks are easily lost or gained!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Carbocation Stability and Markovnikov's Rule
Carbocation stability determines which products form during electrophilic addition. Alkyl groups are electron-releasing, which spreads out the positive charge and stabilises the carbocation.
Tertiary carbocations (3°) are most stable because they have three alkyl groups spreading the charge. Secondary (2°) are less stable, whilst primary (1°) carbocations are least stable.
The major product always forms via the more stable carbocation intermediate. This explains Markovnikov's rule - addition occurs to give the most stable carbocation possible.
Understanding this concept helps predict products in exam questions. Look for which carbocation would be more stable, and you'll know which product predominates.
Quick Check: Count the alkyl groups attached to the positive carbon - more groups mean more stability!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Markovnikov's Rule in Action
This page shows Markovnikov's rule working with a specific example. When HCl adds to an unsymmetrical alkene, two different carbocations can form, but one is much more stable.
The secondary carbocation forms the major product because it's more stable than the primary carbocation. This isn't a 50:50 split - the major product dominates significantly.
In exam questions, you'll need to identify which carbocation is more stable and predict the major product accordingly. Remember that the hydrogen always adds to create the more stable carbocation.
Exam Strategy: When predicting products, always consider both possible carbocations and choose the more stable pathway!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
E/Z Isomerism Basics
E/Z isomerism is a type of stereoisomerism where molecules have the same structural formula but different spatial arrangements. This only occurs when different atoms or groups attach to each carbon of a C=C double bond.
The restricted rotation around double bonds means these different arrangements are actually separate compounds with different properties. Both carbons of the C=C must have different groups attached - if any carbon has two identical groups, no stereoisomerism exists.
Naming stereoisomers uses priority rules based on atomic number. Higher atomic numbers get higher priority, and the arrangement of high-priority groups determines the name.
Z isomers have the highest priority groups on the same side of the double bond, whilst E isomers have them on opposite sides.
Memory Aid: Think "Z = Zame zide" and "E = opposite Ends" to remember the naming system!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Cis-Trans Isomerism and Bonding
Cis-trans isomerism is a special case of E/Z isomerism where two identical groups attach to each carbon of the C=C. Cis means identical groups are on the same side, whilst trans means they're on opposite sides.
The C=C double bond contains both a sigma bond and a pi bond. Sigma bonds form from direct end-to-end orbital overlap, whilst pi bonds form from sideways overlap of p-orbitals above and below the bonding atoms.
Each carbon in C=C makes three sigma bonds using sp² hybridisation. The fourth electron occupies a p-orbital, and these p-orbitals overlap sideways to form the pi bond.
This bonding arrangement prevents rotation around the double bond, which is why stereoisomerism exists in alkenes but not alkanes.
Key Concept: The pi bond's sideways overlap prevents rotation - this restriction is what makes stereoisomerism possible!
We thought you’d never ask...
What is the Knowunity AI companion?
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
Where can I download the Knowunity app?
You can download the app from Google Play Store and Apple App Store.
Is Knowunity really free of charge?
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
Similar content
Most popular content: Organic Chemistry
9A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
Comprehensive Chemistry Overview
Master key chemistry concepts with this detailed cheat sheet covering topics like reaction rates, redox reactions, chromatography, and the periodic table. Perfect for exam preparation, this resource includes essential information on enthalpy changes, organic synthesis, and more. Ideal for A-Level students seeking to enhance their understanding and performance in chemistry.
Organic Chemistry Essentials
Explore key concepts in organic chemistry, including functional groups, IUPAC nomenclature, reaction mechanisms, and polymer chemistry. This comprehensive summary covers aromatic compounds, alkenes, aldehydes, ketones, and mass spectrometry, providing essential insights for students. Ideal for exam preparation and understanding organic synthesis, chemical reactions, and molecular structures.
chemistry spec topics checklist
ocr a level chemistry checklist made from the official specification
OCR A-Level Chemistry Vocabulary
Explore an extensive collection of key terms and concepts for OCR A-Level Chemistry, organized by topic and sub-topic. This resource is designed to enhance your understanding and application of advanced vocabulary in exams, covering essential areas such as atomic structure, organic chemistry, and reaction mechanisms.
Nat 5 chemistry unit 2
All necessary information for national 5 chemistry
Organic chemistry GCSE AQA
GCSE triple chemistry notes
Organic Reaction Mechanisms
Explore key organic reaction mechanisms including electrophilic addition, free radical substitution, and nucleophilic substitution. This summary covers hydrocarbons, alkanes, alkenes, and their transformations, essential for AQA AS Level organic chemistry. Ideal for students seeking to understand complex organic processes and improve exam performance.
Alkanes and Hydrocarbons Overview
Explore the fundamentals of alkanes and hydrocarbons in this concise guide. Understand the molecular and structural formulas, naming conventions, and the significance of isomers. Ideal for Year 12 students preparing for A-Level Organic Chemistry (OCR B Salters). Key concepts include saturated hydrocarbons, IUPAC nomenclature, and the characteristics of alkanes.
Most popular content in Chemistry
9GCSE AQA Chemistry Topic 1- The Atom
The Atom
chem paper 1
higher
A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
GCSE AQA Chemistry Topic 1
Quiz covering all of topic 1
Essential Chemistry Practicals
Explore key AQA GCSE Chemistry practicals, including flame tests, titration, and gas identification. This resource covers essential techniques for analyzing ions, making salts, and understanding reaction kinetics. Perfect for students preparing for exams and practical assessments.
chem paper 1
Unlock the secrets of chemical reactions with this comprehensive flashcard set designed to help you conquer complex concepts and ace your Chemistry exams.
AQA GCSE Chemistry 4.2 Bonding, structure and the Properties of Matter
4.2 Metals and Alloys
AQA GCSE Chemistry 4.4 Chemical changes
4.4 Reactions of acids with bases or carbonates
Fundamentals of Atomic Structure
Practice identifying subatomic particles, calculating neutrons, and defining isotopes and atomic/mass numbers.
Most popular content
9Sociology of Education Overview
Explore comprehensive A-Level Sociology notes on the education system, covering key theories, policies, and sociological perspectives. This resource includes insights on marketisation, gender roles, cultural deprivation, and educational inequalities, providing a thorough understanding of how education shapes social stratification and individual achievement. Ideal for exam preparation and in-depth study.
Sociology of Families: Comprehensive Revision
Dive into an extensive overview of family dynamics, perspectives, and patterns in sociology. This resource covers key concepts such as family diversity, gender roles, marriage, and the impact of social policies on family structures. Perfect for A-Level Sociology students preparing for Paper 2.
Criminology: Crime & Punishment Overview
Comprehensive mindmaps covering key concepts in the Crime and Punishment topic for WJEC Criminology Unit 4. This resource includes detailed insights into the Criminal Justice System, crime prevention strategies, sentencing models, and the roles of various agencies. Ideal for A-Level revision, ensuring you grasp essential theories and legislative processes to excel in your exams.
An Inspector Calls: Character Insights
Explore in-depth analysis and key quotes for characters in J.B. Priestley's 'An Inspector Calls'. This resource covers Gerald Croft, Inspector Goole, Sheila Birling, Mrs. Birling, Eric Birling, and Eva Smith, focusing on themes of class, gender roles, and social responsibility. Ideal for students aiming for Grade 8 and above.
WJEC Unit 4 Criminology
Criminology unit 4 detailed revision note
Criminology Theories Overview
Explore key criminology theories and their implications on crime and deviance. This comprehensive summary covers biological, psychological, and sociological perspectives, including labelling theory, right realism, and the impact of social campaigns on policy development. Ideal for A-Level criminology students seeking to understand the complexities of criminal behaviour and the factors influencing crime prevention strategies.
Romeo and Juliet: Key themes
Key Romeo and Juliet themes and analysed quotes
Macbeth: Guilt and Ambition
Explore the complex themes of guilt and ambition in Shakespeare's 'Macbeth'. This analysis covers key characters, including Macbeth and Lady Macbeth, their moral dilemmas, and the tragic consequences of their ambition. Ideal for students studying character motivations, thematic elements, and the psychological impact of power. Includes insights on the natural order, manipulation, and the descent into madness.
Cell Biology and Cell structure
cell structures
Can't find what you're looking for? Explore other subjects.
Students love us — and so will you.
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.