Understanding atomic structure is fundamental to chemistry - it's like... Show more
Sign up to see the contentIt's free!
Access to all documents
Improve your grades
Join milions of students
Knowunity AI
Subjects
Triangle Congruence and Similarity Theorems
Triangle Properties and Classification
Linear Equations and Graphs
Geometric Angle Relationships
Trigonometric Functions and Identities
Equation Solving Techniques
Circle Geometry Fundamentals
Division Operations and Methods
Basic Differentiation Rules
Exponent and Logarithm Properties
Show all topics
Human Organ Systems
Reproductive Cell Cycles
Biological Sciences Subdisciplines
Cellular Energy Metabolism
Autotrophic Energy Processes
Inheritance Patterns and Principles
Biomolecular Structure and Organization
Cell Cycle and Division Mechanics
Cellular Organization and Development
Biological Structural Organization
Show all topics
Chemical Sciences and Applications
Atomic Structure and Composition
Molecular Electron Structure Representation
Atomic Electron Behavior
Matter Properties and Water
Mole Concept and Calculations
Gas Laws and Behavior
Periodic Table Organization
Chemical Thermodynamics Fundamentals
Chemical Bond Types and Properties
Show all topics
European Renaissance and Enlightenment
European Cultural Movements 800-1920
American Revolution Era 1763-1797
American Civil War 1861-1865
Global Imperial Systems
Mongol and Chinese Dynasties
U.S. Presidents and World Leaders
Historical Sources and Documentation
World Wars Era and Impact
World Religious Systems
Show all topics
Classic and Contemporary Novels
Literary Character Analysis
Rhetorical Theory and Practice
Classic Literary Narratives
Reading Analysis and Interpretation
Narrative Structure and Techniques
English Language Components
Influential English-Language Authors
Basic Sentence Structure
Narrative Voice and Perspective
Show all topics
269
•
Updated Mar 14, 2026
•
jayne
@k.jayn3
Understanding atomic structure is fundamental to chemistry - it's like... Show more










Ever wondered what makes up everything around you? Atoms are the building blocks of matter, and understanding their structure is your gateway to mastering chemistry.
An atom consists of a nucleus at the centre containing protons (positively charged) and neutrons (neutral charge). These particles together are called nucleons, held together by the strong nuclear force - much stronger than the forces between protons and electrons.
Electrons (negatively charged) orbit the nucleus in outer shells. Despite being crucial for chemical reactions, electrons are incredibly light compared to protons and neutrons - about 1,800 times lighter!
Quick Tip: Think of an atom like a solar system - the nucleus is the sun, and electrons are planets orbiting around it.

The atomic number is like an element's fingerprint - it tells you exactly which element you're dealing with. Change this number, and you've got a completely different element. The atomic number equals the number of protons in an atom.
The mass number tells you how many nucleons are in the nucleus. Since electrons are so light, they don't contribute significantly to an atom's mass.
Looking at the data table, you'll see that protons and neutrons have nearly identical masses, whilst electrons are thousands of times lighter. This is why we can ignore electron mass when calculating atomic mass.
Exam Focus: Remember that atomic number = protons, and mass number = protons + neutrons. You'll use this constantly!

Isotopes are atoms of the same element with identical numbers of protons but different numbers of neutrons. They behave the same chemically but have different physical properties like melting points. Some larger isotopes, like Carbon-14, are unstable and radioactive.
Relative atomic mass compares an atom's mass to one-twelfth of a Carbon-12 atom's mass. Carbon-12 is our reference standard because it makes up 98.93% of all carbon isotopes.
To calculate relative atomic mass from isotope data, use: (Isotope 1 × % abundance) + (Isotope 2 × % abundance) ÷ 100. For magnesium's isotopes: (24×79%) + (25×10%) + (26×11%) ÷ 100.
Calculator Ready: These calculations appear frequently in exams, so practice with different isotope mixtures until it becomes automatic.

Mass spectrometry is like a high-tech weighing scale that can identify atoms and molecules by their mass. It works through four main stages that separate particles by how fast they travel.
Ionisation happens in two ways. Electron impact fires high-energy electrons at samples, knocking off electrons to create positive ions . Electrospray ionisation is gentler, used for large molecules like proteins - the sample gains a proton instead .
Acceleration gives all ions the same kinetic energy using an electric field. Since KE = ½mv², lighter ions move faster than heavier ones with the same energy. Detection occurs when ions hit a negatively charged plate, creating a current - more ions mean bigger current.
Real World: This technique is used everywhere from drug testing to identifying explosives at airports!

The beauty of mass spectrometry lies in its ability to identify unknown substances and determine their abundance. You'll need to understand both ionisation methods for your exams.
Electron impact works by bombarding samples with electrons from an electron gun. This aggressive method suits elements and small compounds, creating positive ions by removing electrons completely.
Electrospray ionisation takes a gentler approach for fragile, large molecules. The sample dissolves in a volatile solvent and gets sprayed through a charged needle, picking up protons to become positive ions.
After acceleration, all ions have identical kinetic energy. Using KE = ½mv², you can see why lighter ions travel faster - they need higher velocity to match the kinetic energy of heavier ions.
Exam Strategy: Practice identifying which ionisation method suits different types of compounds - it's a common exam question!

In the ion drift stage, ions travel through a vacuum tube at different speeds based on their mass. The vacuum prevents unwanted collisions with air particles that would mess up your results.
Detection is brilliantly simple - positive ions hit a detector plate and grab electrons. This creates electron flow (current) as electrons move to replace the missing ones. More ions hitting the detector means greater current, showing higher abundance.
The time of flight calculations use the relationship between kinetic energy and velocity . Since all ions have the same kinetic energy, lighter ions must travel faster to reach the detector first.
Maths Connection: These calculations combine physics equations you already know - kinetic energy and velocity formulas working together.

Forget GCSE electron shells - A-level uses energy levels divided into sub-levels (s, p, d, f). Think of it like a hotel with floors (energy levels) and different room types holding specific numbers of guests (electrons).
Sub-level capacity is crucial: s holds 2 electrons, p holds 6, d holds 10, and f holds 14. Electrons live in orbitals - regions of space around the nucleus.
Writing electron configuration shows where electrons live. Oxygen (8 electrons) is 1s²2s²2p⁴. Since oxygen's outermost electrons occupy p-orbitals, it's a p-block element - matching its position on the periodic table.
Memory Trick: Remember "2, 6, 10, 14" for s, p, d, f capacities - it's the foundation for all electron configurations.

Noble gas shorthand saves time by using the nearest noble gas as a starting point. Instead of writing magnesium as 1s²2s²2p⁶3s², you can write [Ne]3s² - much quicker for larger atoms!
Electron spin follows important rules. Electrons in the same orbital must have opposite spins for stability. When filling orbitals, electrons prefer to occupy empty orbitals first before pairing up - like people preferring empty bus seats before sharing.
The energy level diagram shows how orbitals fill in order. Notice that 4s fills before 3d - this unusual order catches many students out in exams.
Exam Trap: Remember 4s fills before 3d, but when writing electron configurations for ions, remove 4s electrons first!

Ionisation energy is the energy needed to remove an electron from a gaseous atom. Think of it as the "stickiness" between the nucleus and electrons - stronger attraction means higher ionisation energy.
Successive ionisation energies increase dramatically. Removing the first electron from sodium takes 496 kJ/mol, but the second needs 4563 kJ/mol! This happens because removing electrons increases the positive charge, creating stronger attraction to remaining electrons.
Three factors affect ionisation energy: atomic radius (bigger atoms hold electrons more weakly), nuclear charge , and shielding (inner electrons repel outer ones).
Pattern Recognition: Massive jumps in successive ionisation energies reveal electron shell structures - perfect for identifying group numbers!
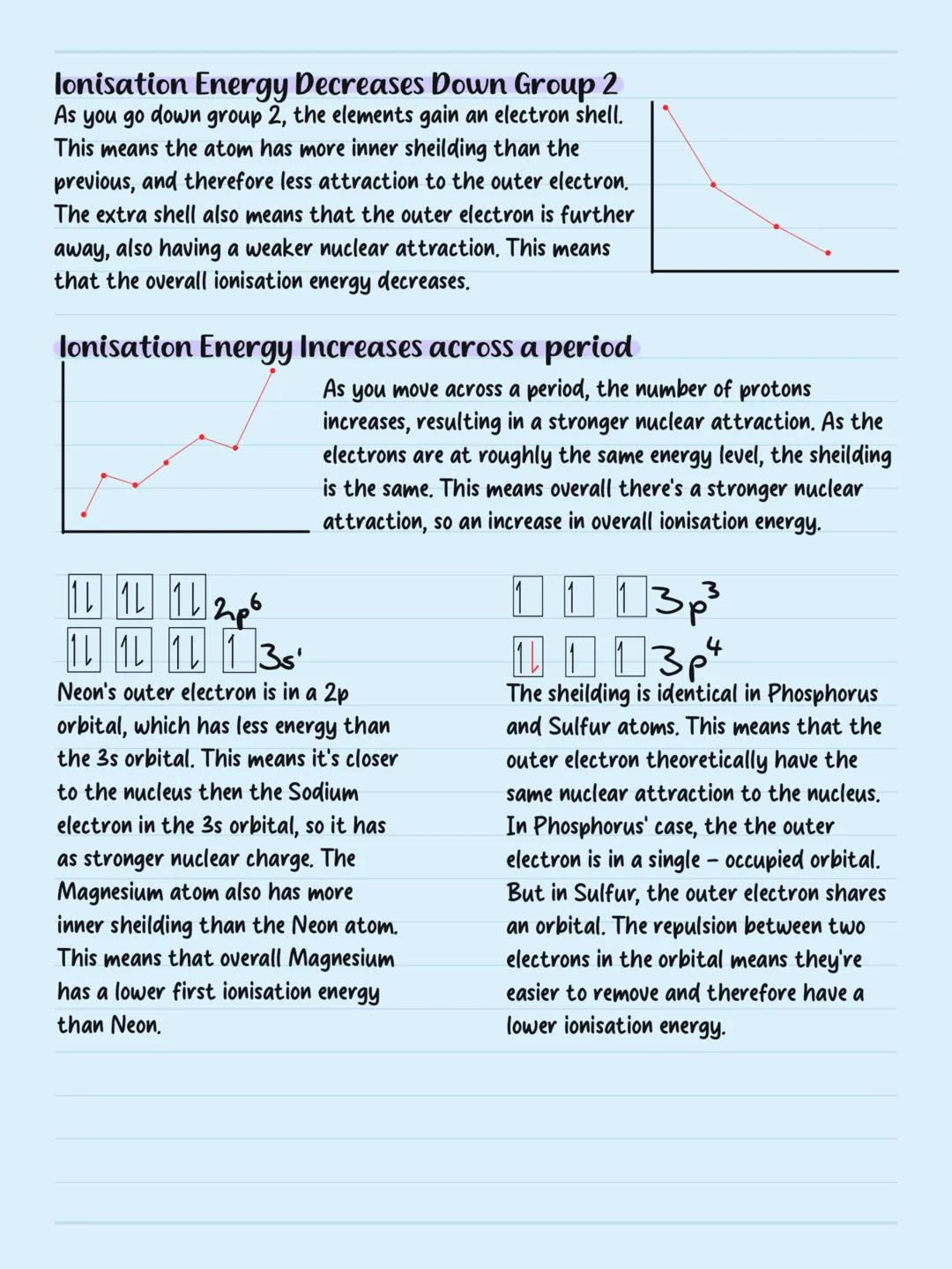
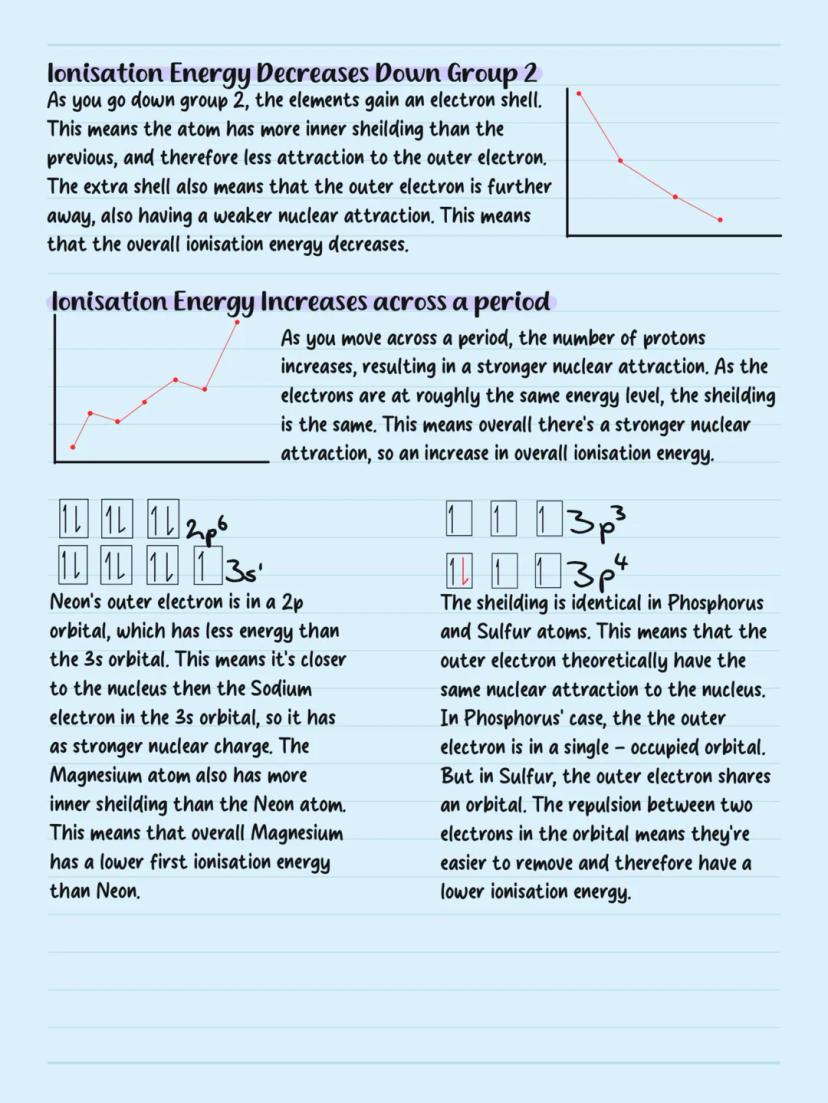
Ionisation energy decreases down groups because atoms get larger and gain more electron shells. The outer electrons are further from the nucleus and experience more shielding from inner electrons, making them easier to remove.
Ionisation energy increases across periods as nuclear charge increases while electrons stay at roughly the same distance. More protons mean stronger attraction to outer electrons.
Some interesting exceptions exist: magnesium has lower first ionisation energy than neon because magnesium's outer electron is in a higher energy 3s orbital. Sulfur has lower ionisation energy than phosphorus due to electron repulsion - sulfur's outer electron shares an orbital, making it easier to remove.
Trend Breakers: Learn the exceptions in periods 2 and 3 - they're exam favourites and show you understand electron-electron repulsion!
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
You can download the app from Google Play Store and Apple App Store.
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
App Store
Google Play
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
jayne
@k.jayn3
Understanding atomic structure is fundamental to chemistry - it's like learning the alphabet before you can read. These notes cover everything from the basic parts of atoms to advanced concepts like mass spectrometry and ionisation energy that you'll need for... Show more

Access to all documents
Improve your grades
Join milions of students
Ever wondered what makes up everything around you? Atoms are the building blocks of matter, and understanding their structure is your gateway to mastering chemistry.
An atom consists of a nucleus at the centre containing protons (positively charged) and neutrons (neutral charge). These particles together are called nucleons, held together by the strong nuclear force - much stronger than the forces between protons and electrons.
Electrons (negatively charged) orbit the nucleus in outer shells. Despite being crucial for chemical reactions, electrons are incredibly light compared to protons and neutrons - about 1,800 times lighter!
Quick Tip: Think of an atom like a solar system - the nucleus is the sun, and electrons are planets orbiting around it.

Access to all documents
Improve your grades
Join milions of students
The atomic number is like an element's fingerprint - it tells you exactly which element you're dealing with. Change this number, and you've got a completely different element. The atomic number equals the number of protons in an atom.
The mass number tells you how many nucleons are in the nucleus. Since electrons are so light, they don't contribute significantly to an atom's mass.
Looking at the data table, you'll see that protons and neutrons have nearly identical masses, whilst electrons are thousands of times lighter. This is why we can ignore electron mass when calculating atomic mass.
Exam Focus: Remember that atomic number = protons, and mass number = protons + neutrons. You'll use this constantly!

Access to all documents
Improve your grades
Join milions of students
Isotopes are atoms of the same element with identical numbers of protons but different numbers of neutrons. They behave the same chemically but have different physical properties like melting points. Some larger isotopes, like Carbon-14, are unstable and radioactive.
Relative atomic mass compares an atom's mass to one-twelfth of a Carbon-12 atom's mass. Carbon-12 is our reference standard because it makes up 98.93% of all carbon isotopes.
To calculate relative atomic mass from isotope data, use: (Isotope 1 × % abundance) + (Isotope 2 × % abundance) ÷ 100. For magnesium's isotopes: (24×79%) + (25×10%) + (26×11%) ÷ 100.
Calculator Ready: These calculations appear frequently in exams, so practice with different isotope mixtures until it becomes automatic.

Access to all documents
Improve your grades
Join milions of students
Mass spectrometry is like a high-tech weighing scale that can identify atoms and molecules by their mass. It works through four main stages that separate particles by how fast they travel.
Ionisation happens in two ways. Electron impact fires high-energy electrons at samples, knocking off electrons to create positive ions . Electrospray ionisation is gentler, used for large molecules like proteins - the sample gains a proton instead .
Acceleration gives all ions the same kinetic energy using an electric field. Since KE = ½mv², lighter ions move faster than heavier ones with the same energy. Detection occurs when ions hit a negatively charged plate, creating a current - more ions mean bigger current.
Real World: This technique is used everywhere from drug testing to identifying explosives at airports!

Access to all documents
Improve your grades
Join milions of students
The beauty of mass spectrometry lies in its ability to identify unknown substances and determine their abundance. You'll need to understand both ionisation methods for your exams.
Electron impact works by bombarding samples with electrons from an electron gun. This aggressive method suits elements and small compounds, creating positive ions by removing electrons completely.
Electrospray ionisation takes a gentler approach for fragile, large molecules. The sample dissolves in a volatile solvent and gets sprayed through a charged needle, picking up protons to become positive ions.
After acceleration, all ions have identical kinetic energy. Using KE = ½mv², you can see why lighter ions travel faster - they need higher velocity to match the kinetic energy of heavier ions.
Exam Strategy: Practice identifying which ionisation method suits different types of compounds - it's a common exam question!

Access to all documents
Improve your grades
Join milions of students
In the ion drift stage, ions travel through a vacuum tube at different speeds based on their mass. The vacuum prevents unwanted collisions with air particles that would mess up your results.
Detection is brilliantly simple - positive ions hit a detector plate and grab electrons. This creates electron flow (current) as electrons move to replace the missing ones. More ions hitting the detector means greater current, showing higher abundance.
The time of flight calculations use the relationship between kinetic energy and velocity . Since all ions have the same kinetic energy, lighter ions must travel faster to reach the detector first.
Maths Connection: These calculations combine physics equations you already know - kinetic energy and velocity formulas working together.

Access to all documents
Improve your grades
Join milions of students
Forget GCSE electron shells - A-level uses energy levels divided into sub-levels (s, p, d, f). Think of it like a hotel with floors (energy levels) and different room types holding specific numbers of guests (electrons).
Sub-level capacity is crucial: s holds 2 electrons, p holds 6, d holds 10, and f holds 14. Electrons live in orbitals - regions of space around the nucleus.
Writing electron configuration shows where electrons live. Oxygen (8 electrons) is 1s²2s²2p⁴. Since oxygen's outermost electrons occupy p-orbitals, it's a p-block element - matching its position on the periodic table.
Memory Trick: Remember "2, 6, 10, 14" for s, p, d, f capacities - it's the foundation for all electron configurations.

Access to all documents
Improve your grades
Join milions of students
Noble gas shorthand saves time by using the nearest noble gas as a starting point. Instead of writing magnesium as 1s²2s²2p⁶3s², you can write [Ne]3s² - much quicker for larger atoms!
Electron spin follows important rules. Electrons in the same orbital must have opposite spins for stability. When filling orbitals, electrons prefer to occupy empty orbitals first before pairing up - like people preferring empty bus seats before sharing.
The energy level diagram shows how orbitals fill in order. Notice that 4s fills before 3d - this unusual order catches many students out in exams.
Exam Trap: Remember 4s fills before 3d, but when writing electron configurations for ions, remove 4s electrons first!

Access to all documents
Improve your grades
Join milions of students
Ionisation energy is the energy needed to remove an electron from a gaseous atom. Think of it as the "stickiness" between the nucleus and electrons - stronger attraction means higher ionisation energy.
Successive ionisation energies increase dramatically. Removing the first electron from sodium takes 496 kJ/mol, but the second needs 4563 kJ/mol! This happens because removing electrons increases the positive charge, creating stronger attraction to remaining electrons.
Three factors affect ionisation energy: atomic radius (bigger atoms hold electrons more weakly), nuclear charge , and shielding (inner electrons repel outer ones).
Pattern Recognition: Massive jumps in successive ionisation energies reveal electron shell structures - perfect for identifying group numbers!

Access to all documents
Improve your grades
Join milions of students
Ionisation energy decreases down groups because atoms get larger and gain more electron shells. The outer electrons are further from the nucleus and experience more shielding from inner electrons, making them easier to remove.
Ionisation energy increases across periods as nuclear charge increases while electrons stay at roughly the same distance. More protons mean stronger attraction to outer electrons.
Some interesting exceptions exist: magnesium has lower first ionisation energy than neon because magnesium's outer electron is in a higher energy 3s orbital. Sulfur has lower ionisation energy than phosphorus due to electron repulsion - sulfur's outer electron shares an orbital, making it easier to remove.
Trend Breakers: Learn the exceptions in periods 2 and 3 - they're exam favourites and show you understand electron-electron repulsion!
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
You can download the app from Google Play Store and Apple App Store.
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
9
Smart Tools NEW
Transform this note into: ✓ 50+ Practice Questions ✓ Interactive Flashcards ✓ Full Mock Exam ✓ Essay Outlines
Explore the fundamental concepts of atomic structure, including protons, neutrons, and electrons. This summary covers the composition of atoms, their charges, and how ions are formed. Ideal for AQA A Level Chemistry students seeking a clear understanding of atomic theory.
Explore the principles and methods of mass spectrometry, including electron impact and electrospray ionisation. Understand how molecular ions are formed, the significance of mass spectra, and how to calculate relative atomic mass. Ideal for AQA A-level chemistry students.
Explore the concepts of atom economy, percentage yield, and the significance of hydrated vs. anhydrous salts in A-level Physical Chemistry. This summary covers key calculations, the ideal gas equation, and the importance of efficient chemical reactions for sustainability. Ideal for students preparing for exams.
Explore the fundamentals of atomic structure, including ionization energies, electron configurations, isotopes, and mass spectrometry. This comprehensive summary covers key concepts such as atomic radius, shielding effects, and the historical development of atomic models. Ideal for A-level Chemistry students preparing for exams.
Explore the fundamentals of isotopes, including definitions, calculations of relative atomic mass, and the relationship between protons, neutrons, and mass number. This summary covers key concepts such as relative abundance and atomic structure, essential for mastering Module 2 in Chemistry.
Explore the fundamentals of atomic structure, relative atomic mass, and isotopes in this detailed summary. Understand concepts like average atomic mass, ionization, and the role of mass spectrometry in determining percentage abundance. Ideal for A Level Chemistry students preparing for exams.
App Store
Google Play
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user