Amines are nitrogen-containing organic compounds that play crucial roles in... Show more
Sign up to see the contentIt's free!
Access to all documents
Improve your grades
Join milions of students
Knowunity AI
Subjects
Triangle Congruence and Similarity Theorems
Triangle Properties and Classification
Linear Equations and Graphs
Geometric Angle Relationships
Trigonometric Functions and Identities
Equation Solving Techniques
Circle Geometry Fundamentals
Division Operations and Methods
Basic Differentiation Rules
Exponent and Logarithm Properties
Show all topics
Human Organ Systems
Reproductive Cell Cycles
Biological Sciences Subdisciplines
Cellular Energy Metabolism
Autotrophic Energy Processes
Inheritance Patterns and Principles
Biomolecular Structure and Organization
Cell Cycle and Division Mechanics
Cellular Organization and Development
Biological Structural Organization
Show all topics
Chemical Sciences and Applications
Atomic Structure and Composition
Molecular Electron Structure Representation
Atomic Electron Behavior
Matter Properties and Water
Mole Concept and Calculations
Gas Laws and Behavior
Periodic Table Organization
Chemical Thermodynamics Fundamentals
Chemical Bond Types and Properties
Show all topics
European Renaissance and Enlightenment
European Cultural Movements 800-1920
American Revolution Era 1763-1797
American Civil War 1861-1865
Global Imperial Systems
Mongol and Chinese Dynasties
U.S. Presidents and World Leaders
Historical Sources and Documentation
World Wars Era and Impact
World Religious Systems
Show all topics
Classic and Contemporary Novels
Literary Character Analysis
Rhetorical Theory and Practice
Classic Literary Narratives
Reading Analysis and Interpretation
Narrative Structure and Techniques
English Language Components
Influential English-Language Authors
Basic Sentence Structure
Narrative Voice and Perspective
Show all topics
170
•
Updated May 11, 2026
•
Zainab
@zainabkaz_dvgr
Amines are nitrogen-containing organic compounds that play crucial roles in... Show more


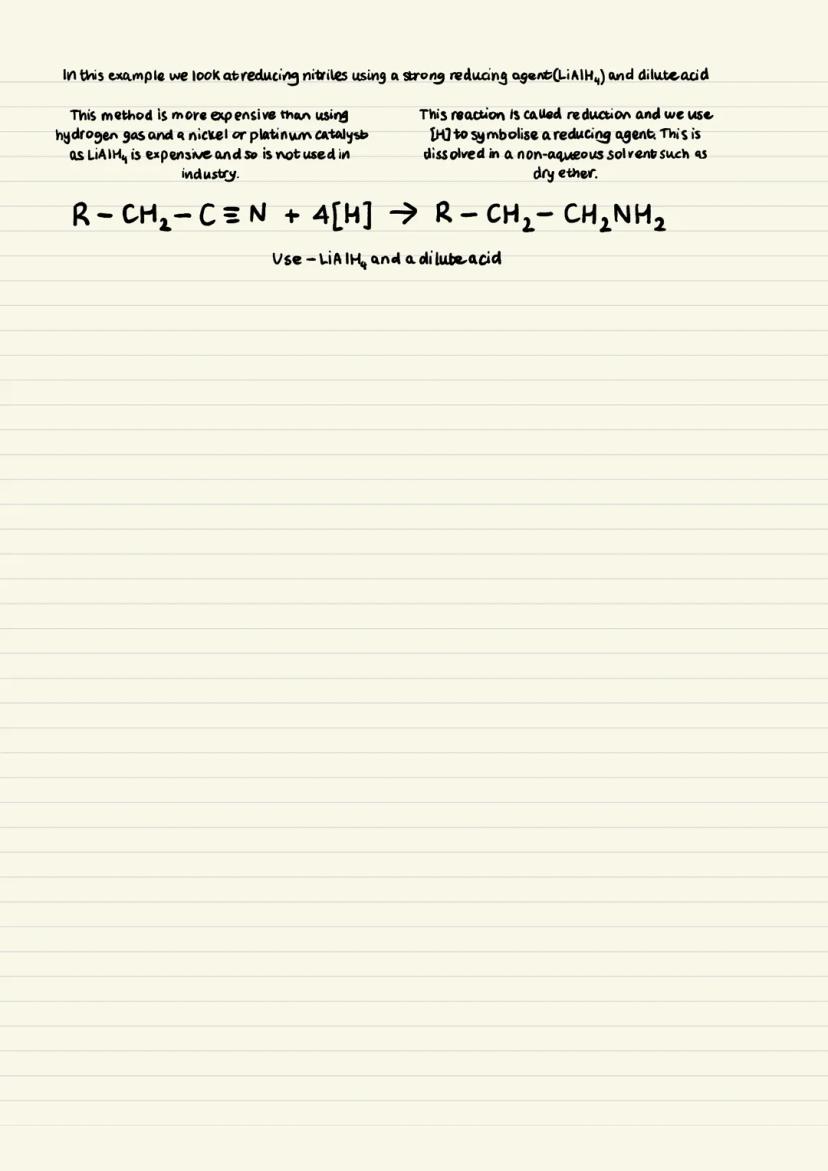
Amines are classified based on how many carbon groups are attached to the nitrogen atom. Primary amines have one carbon group attached (like methylamine), secondary amines have two (like dimethylamine), and tertiary amines have three (like trimethylamine).
When a fourth group bonds to nitrogen, you get a quaternary ammonium salt with a positive charge. This creates compounds like tetramethylammonium, which are important in various chemical processes.
The key difference between aliphatic amines and aromatic amines (containing benzene rings) affects their chemical behaviour significantly. Phenylamine is the most common aromatic amine you'll encounter.
Quick Tip: Remember the classification by counting carbon groups attached to nitrogen - it's that simple!

Amines act as bases because they have a lone pair of electrons on nitrogen that can accept protons. When this happens, a dative covalent bond forms where both electrons come from the nitrogen's lone pair.
The strength of an amine as a base depends on how available these lone pair electrons are. Electron density around nitrogen determines this availability - higher density means stronger basicity.
The order of base strength is: Primary aliphatic amines > Ammonia > Aromatic amines. Aromatic amines are the weakest because the benzene ring pulls electrons away from nitrogen through delocalisation. Aliphatic amines are stronger because alkyl groups push electrons towards nitrogen.
This electron availability also makes amines excellent nucleophiles - they readily attack electron-deficient centres in chemical reactions.
Remember: Think of aromatic rings as electron-hungry - they steal electron density from nitrogen, making aromatic amines weaker bases.

There are two main ways to synthesise aliphatic amines in the lab and industry. The first involves reacting halogenoalkanes with excess ammonia.
In this mechanism, ammonia acts as a nucleophile and attacks the carbon attached to the halogen. This forms an alkylammonium salt intermediate with a positively charged nitrogen and a chloride ion.
A second ammonia molecule then removes a hydrogen from this salt, producing the primary amine and ammonium chloride. However, this method has a major drawback - it produces a mixture of primary, secondary, tertiary amines, and quaternary salts.
This happens because the primary amine still has lone pairs and can act as a nucleophile itself, reacting with more halogenoalkane molecules. That's why you get an impure product that needs separation.
Exam Tip: Always mention the impurity problem when discussing the halogenoalkane method - it's a common exam question!
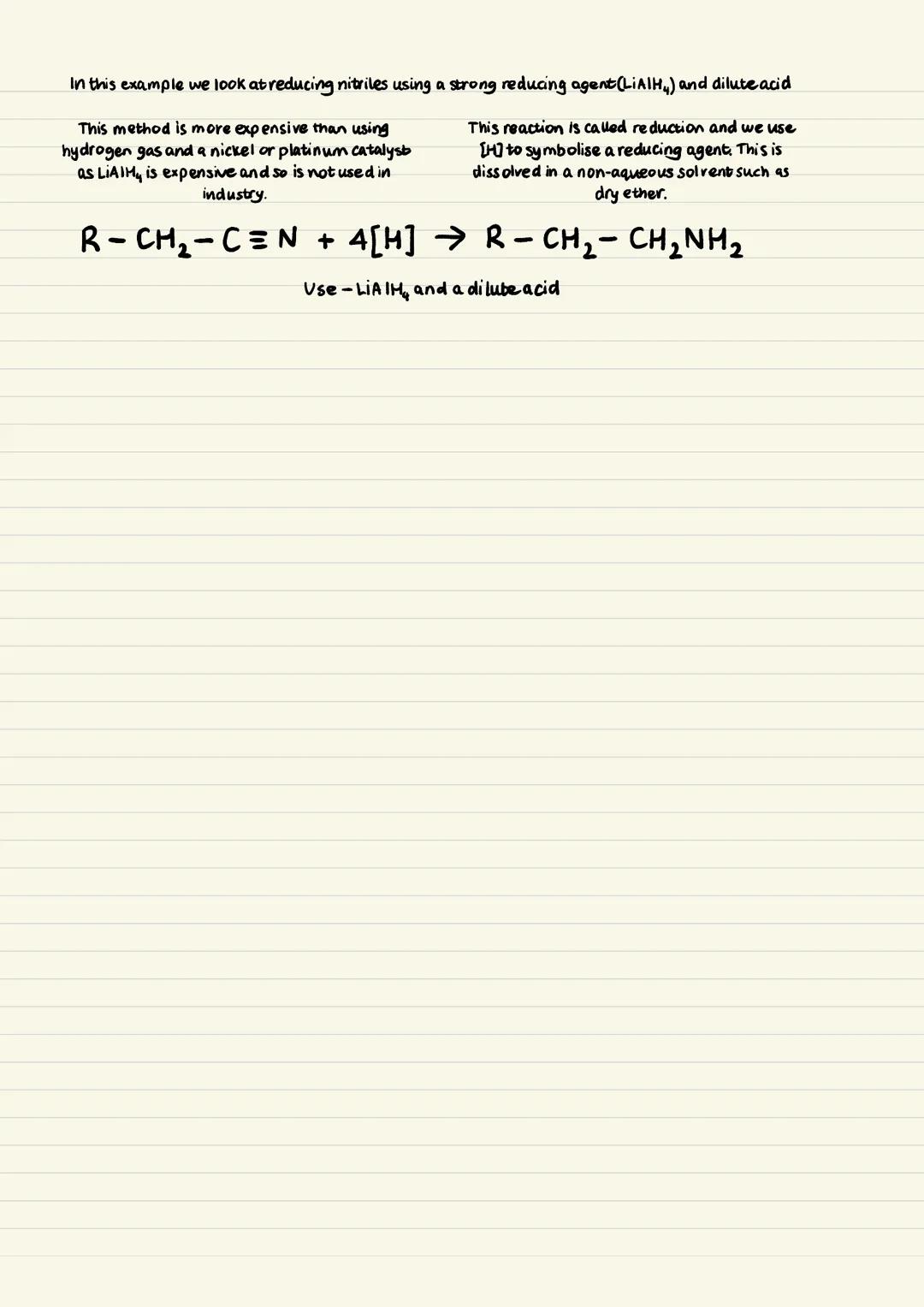
The industrial preference is reducing nitriles using hydrogen gas with a nickel or platinum catalyst. This method produces only primary amines, giving you a pure product - that's why it's favoured commercially.
The reaction requires high temperature and pressure conditions for catalytic hydrogenation. The nitrile group gets completely reduced to form an amine group, with the carbon chain length increasing by one.
An alternative laboratory method uses LiAlH₄ (lithium aluminium hydride) as a reducing agent with dilute acid. This also produces pure primary amines through reduction, symbolised by [H] in equations.
However, the LiAlH₄ method is expensive and requires dry, non-aqueous solvents like ether. That's why industry sticks with the cheaper hydrogen gas method despite the harsher conditions needed.
Cost vs Purity: Remember that industry always considers economics - the hydrogen method wins because it's both pure and cheap!
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
You can download the app from Google Play Store and Apple App Store.
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
App Store
Google Play
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
Zainab
@zainabkaz_dvgr
Amines are nitrogen-containing organic compounds that play crucial roles in chemistry and biology. Understanding their structure, basicity, and synthesis methods is essential for A-level chemistry success.

Access to all documents
Improve your grades
Join milions of students
Amines are classified based on how many carbon groups are attached to the nitrogen atom. Primary amines have one carbon group attached (like methylamine), secondary amines have two (like dimethylamine), and tertiary amines have three (like trimethylamine).
When a fourth group bonds to nitrogen, you get a quaternary ammonium salt with a positive charge. This creates compounds like tetramethylammonium, which are important in various chemical processes.
The key difference between aliphatic amines and aromatic amines (containing benzene rings) affects their chemical behaviour significantly. Phenylamine is the most common aromatic amine you'll encounter.
Quick Tip: Remember the classification by counting carbon groups attached to nitrogen - it's that simple!

Access to all documents
Improve your grades
Join milions of students
Amines act as bases because they have a lone pair of electrons on nitrogen that can accept protons. When this happens, a dative covalent bond forms where both electrons come from the nitrogen's lone pair.
The strength of an amine as a base depends on how available these lone pair electrons are. Electron density around nitrogen determines this availability - higher density means stronger basicity.
The order of base strength is: Primary aliphatic amines > Ammonia > Aromatic amines. Aromatic amines are the weakest because the benzene ring pulls electrons away from nitrogen through delocalisation. Aliphatic amines are stronger because alkyl groups push electrons towards nitrogen.
This electron availability also makes amines excellent nucleophiles - they readily attack electron-deficient centres in chemical reactions.
Remember: Think of aromatic rings as electron-hungry - they steal electron density from nitrogen, making aromatic amines weaker bases.

Access to all documents
Improve your grades
Join milions of students
There are two main ways to synthesise aliphatic amines in the lab and industry. The first involves reacting halogenoalkanes with excess ammonia.
In this mechanism, ammonia acts as a nucleophile and attacks the carbon attached to the halogen. This forms an alkylammonium salt intermediate with a positively charged nitrogen and a chloride ion.
A second ammonia molecule then removes a hydrogen from this salt, producing the primary amine and ammonium chloride. However, this method has a major drawback - it produces a mixture of primary, secondary, tertiary amines, and quaternary salts.
This happens because the primary amine still has lone pairs and can act as a nucleophile itself, reacting with more halogenoalkane molecules. That's why you get an impure product that needs separation.
Exam Tip: Always mention the impurity problem when discussing the halogenoalkane method - it's a common exam question!
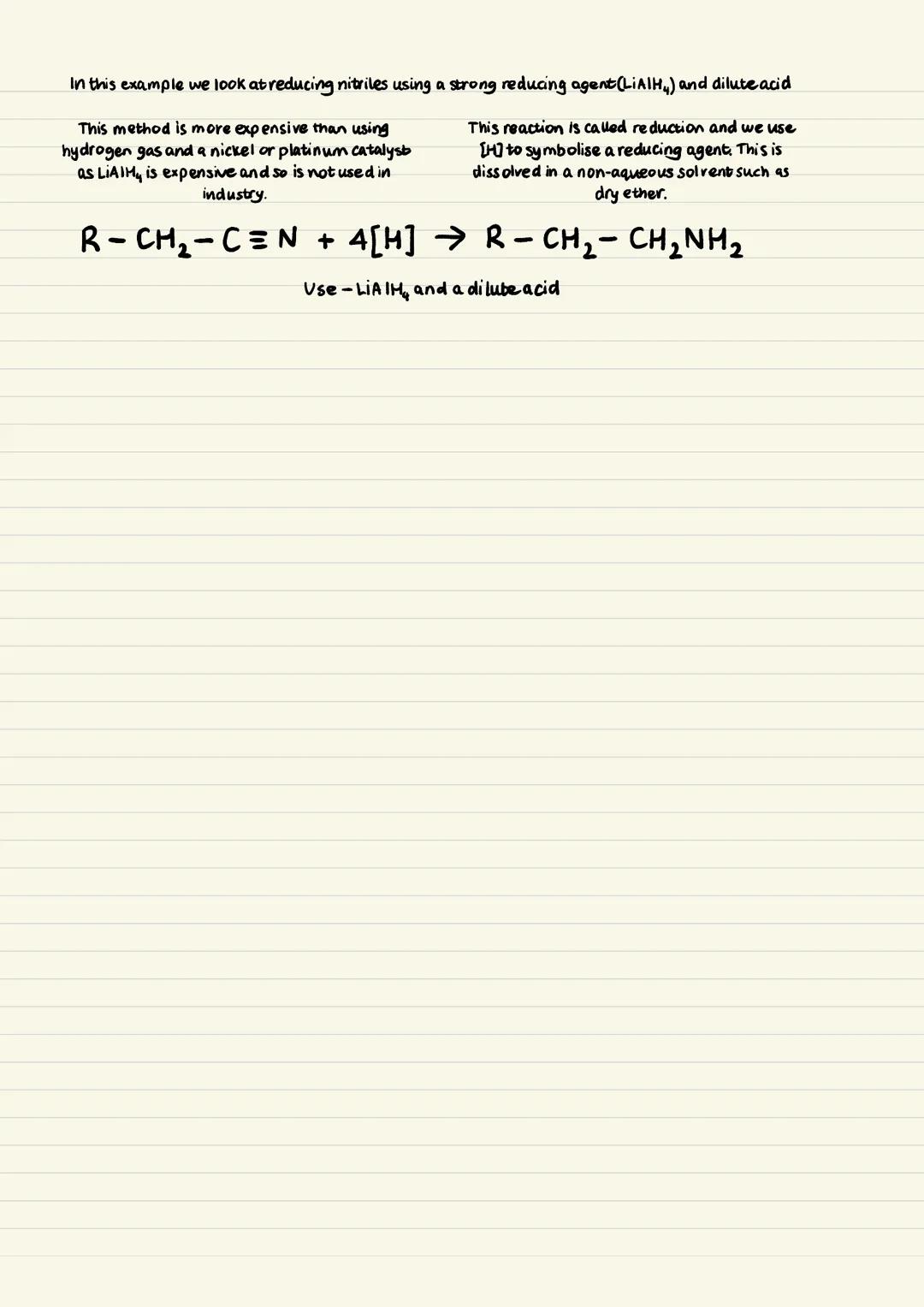
Access to all documents
Improve your grades
Join milions of students
The industrial preference is reducing nitriles using hydrogen gas with a nickel or platinum catalyst. This method produces only primary amines, giving you a pure product - that's why it's favoured commercially.
The reaction requires high temperature and pressure conditions for catalytic hydrogenation. The nitrile group gets completely reduced to form an amine group, with the carbon chain length increasing by one.
An alternative laboratory method uses LiAlH₄ (lithium aluminium hydride) as a reducing agent with dilute acid. This also produces pure primary amines through reduction, symbolised by [H] in equations.
However, the LiAlH₄ method is expensive and requires dry, non-aqueous solvents like ether. That's why industry sticks with the cheaper hydrogen gas method despite the harsher conditions needed.
Cost vs Purity: Remember that industry always considers economics - the hydrogen method wins because it's both pure and cheap!
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
You can download the app from Google Play Store and Apple App Store.
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
7
Smart Tools NEW
Transform this note into: ✓ 50+ Practice Questions ✓ Interactive Flashcards ✓ Full Mock Exam ✓ Essay Outlines
Explore the key concepts of aromatic chemistry, including the structure and reactivity of benzene, electrophilic substitution mechanisms, and the role of delocalized electrons. This summary covers nitration and Friedel-Crafts reactions, comparing the reactivity of alkenes and arenes, and the unique properties of phenols. Ideal for A-Level organic chemistry students.
Explore the mechanisms of nucleophilic substitution in halogenoalkanes, including the formation of alcohols and amines. Understand the role of nucleophiles, the conditions for elimination reactions, and the impact of ozone depletion caused by CFCs. This summary covers key concepts such as halide ions, reaction mechanisms, and radical substitution, essential for AQA AS Chemistry students.
Explore the structure, stability, and electrophilic substitution mechanisms of benzenes and phenols. This summary covers key concepts such as delocalization, resonance, and the differences between electron-donating and electron-withdrawing groups, providing essential insights for A Level Chemistry students.
Explore the mechanisms of halogenoalkanes, focusing on nucleophilic substitution (SN1 and SN2) and elimination reactions. This summary covers key concepts such as the formation of halogenoalkanes, the role of halide ions, and the conditions influencing reaction pathways. Ideal for CIE revision.
Explore key concepts in isomerism and carbonyl compounds, including optical activity, aldehydes, ketones, and esters. This comprehensive A-level chemistry resource covers functional groups, separation methods, and laboratory techniques essential for organic synthesis. Ideal for AQA students preparing for exams.
Explore the reactivity and mechanisms of benzene and phenol, including electrophilic substitution reactions, delocalization of electrons, and key comparisons between alkenes and arenes. This summary covers essential concepts such as nitration, halogenation, and Friedel-Crafts reactions, providing a comprehensive understanding for students studying aromatic compounds.
App Store
Google Play
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
Stefan S
iOS user
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Samantha Klich
Android user
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Anna
iOS user
Best app on earth! no words because it’s too good
Thomas R
iOS user
Just amazing. Let's me revise 10x better, this app is a quick 10/10. I highly recommend it to anyone. I can watch and search for notes. I can save them in the subject folder. I can revise it any time when I come back. If you haven't tried this app, you're really missing out.
Basil
Android user
This app has made me feel so much more confident in my exam prep, not only through boosting my own self confidence through the features that allow you to connect with others and feel less alone, but also through the way the app itself is centred around making you feel better. It is easy to navigate, fun to use, and helpful to anyone struggling in absolutely any way.
David K
iOS user
The app's just great! All I have to do is enter the topic in the search bar and I get the response real fast. I don't have to watch 10 YouTube videos to understand something, so I'm saving my time. Highly recommended!
Sudenaz Ocak
Android user
In school I was really bad at maths but thanks to the app, I am doing better now. I am so grateful that you made the app.
Greenlight Bonnie
Android user
very reliable app to help and grow your ideas of Maths, English and other related topics in your works. please use this app if your struggling in areas, this app is key for that. wish I'd of done a review before. and it's also free so don't worry about that.
Rohan U
Android user
I know a lot of apps use fake accounts to boost their reviews but this app deserves it all. Originally I was getting 4 in my English exams and this time I got a grade 7. I didn’t even know about this app three days until the exam and it has helped A LOT. Please actually trust me and use it as I’m sure you too will see developments.
Xander S
iOS user
THE QUIZES AND FLASHCARDS ARE SO USEFUL AND I LOVE Knowunity AI. IT ALSO IS LITREALLY LIKE CHATGPT BUT SMARTER!! HELPED ME WITH MY MASCARA PROBLEMS TOO!! AS WELL AS MY REAL SUBJECTS ! DUHHH 😍😁😲🤑💗✨🎀😮
Elisha
iOS user
This apps acc the goat. I find revision so boring but this app makes it so easy to organize it all and then you can ask the freeeee ai to test yourself so good and you can easily upload your own stuff. highly recommend as someone taking mocks now
Paul T
iOS user