Understanding atomic structure is absolutely crucial for mastering chemistry -... Show more
Understanding Atomic Structure and Isotopes for OCR A-Level Chemistry






Atomic Structure Basics
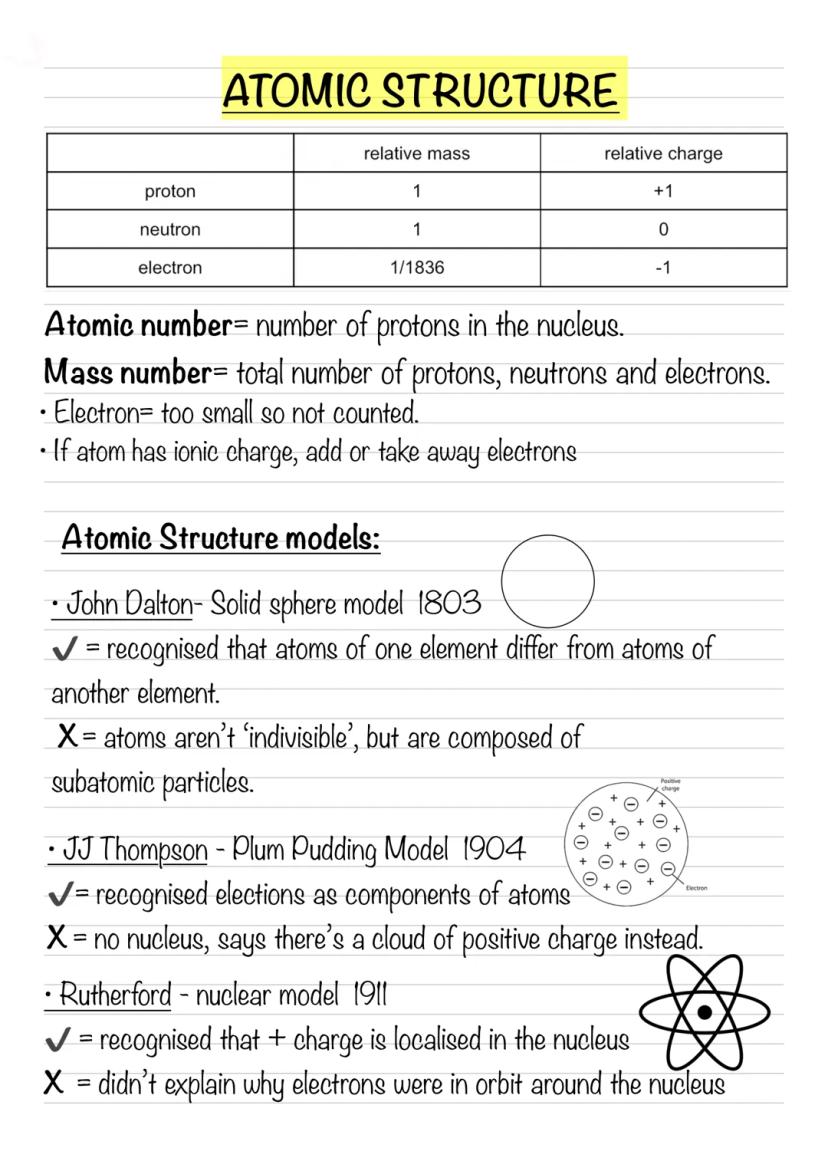
Every atom is made up of three fundamental particles that you need to know inside out. Protons have a relative mass of 1 and a charge of +1, neutrons also have a relative mass of 1 but are neutral (charge of 0), and electrons are tiny with a relative mass of 1/1836 and a charge of -1.
The atomic number tells you how many protons are in the nucleus - this never changes for a given element. The mass number is the total of protons and neutrons (electrons are too light to count). When dealing with ions, simply add or subtract electrons based on the charge.
Key Tip: Remember that protons define what element you're dealing with - change the number of protons, and you've got a completely different element!

Historical Models of the Atom
Scientists didn't always understand atomic structure - it took centuries of brilliant minds to figure it out! John Dalton (1803) imagined atoms as solid spheres, which was groundbreaking but missed the fact that atoms contain smaller particles.
JJ Thompson (1904) discovered electrons and proposed the plum pudding model - electrons embedded in a positive "pudding." Rutherford (1911) proved there's a dense nucleus through his famous gold foil experiment. Niels Bohr (1913) suggested electrons orbit in fixed paths, explaining why atoms don't collapse.
Finally, Schrödinger (1926) gave us the modern quantum model, where electrons exist in probability clouds called orbitals rather than fixed orbits. This model actually works for all atoms and is what we use today.
Remember: Each model built on the previous one's strengths whilst fixing its weaknesses - that's how science progresses!

Isotopes and Relative Mass
Isotopes are atoms of the same element with different numbers of neutrons - they're like identical twins with different weights! They have the same atomic number but different mass numbers, which means they behave almost identically in chemical reactions.
The extra neutrons don't affect chemical properties much, but they do change physical properties like melting points and densities. This is why hydrogen and deuterium (heavy hydrogen) have slightly different boiling points.
Relative atomic mass is the weighted average mass of all an element's isotopes compared to 1/12th of a carbon-12 atom. Relative isotopic mass refers to individual isotopes. These definitions might seem abstract, but they're essential for understanding why atomic masses aren't whole numbers.
Quick Check: If you see a non-whole atomic mass on the periodic table, you know that element has multiple isotopes!

Mass Spectrometry - The Basics
Mass spectrometry is like a sophisticated weighing scale that can identify and measure individual atoms and molecules. It tells you exactly what isotopes are present and in what percentages - incredibly useful for chemists!
The process starts by bombarding your sample with high-energy electrons, which knocks electrons off atoms to create positive ions. These ions get accelerated through an electric field, then separated based on their mass-to-charge ratio .
Since most ions have a +1 charge, the m/z ratio essentially just tells you the mass. A computer detects these separated ions and creates a mass spectrum - a graph showing what masses are present and how abundant they are.
Real-world Application: Mass spectrometry is used everywhere from detecting drugs in athletes to identifying unknown compounds in forensic investigations!

How Mass Spectrometry Works
There are two main methods for separating ions in mass spectrometry. The magnetic deflection method uses a magnetic field to bend the path of ions - heavier ions get deflected less than lighter ones, allowing separation.
Time of flight spectrometry is more elegant - all ions get the same kinetic energy, so lighter ions move faster than heavier ones. By measuring how long it takes ions to travel a fixed distance, you can calculate their masses with incredible precision.
The final mass spectrum is straightforward to read: the x-axis shows relative isotopic mass, and the y-axis shows the percentage abundance of each isotope. The height of each peak tells you how common that particular isotope is.
Pro Tip: The tallest peak represents the most abundant isotope - this is often called the base peak!

Calculating Relative Atomic Mass
You'll definitely need to master these calculations for your exams! The formula is: Ar = Σ(relative isotopic mass × abundance) ÷ 100. This weighted average accounts for how common each isotope is.
For missing isotope masses, substitute 'x' for the unknown value, expand the brackets, multiply by 100, then solve the linear equation. It's just algebra with a chemistry twist!
When calculating percentage abundances, call one isotope 'x' and the other '100-x', then follow the same process. Your final percentages must always add up to 100% - if they don't, you've made an error somewhere.
Exam Success: Always double-check your percentages add to 100% and that your final atomic mass makes sense compared to the individual isotope masses!

Relative Molecular and Formula Mass
Calculating relative molecular mass and relative formula mass is refreshingly straightforward after all those isotope calculations! Simply add up the relative atomic masses of all atoms in the compound.
Use relative molecular mass for simple molecules (like H₂O or CO₂) and relative formula mass for compounds with giant structures like ionic compounds. The calculation method is identical - just different terminology.
Start by writing out all elements present, look up their relative atomic masses from the periodic table, multiply by how many of each atom you have, then add everything together. That's your final answer!
Time-Saver: Make a systematic list of elements and their quantities before calculating - it prevents silly mistakes and saves time in exams!
We thought you’d never ask...
What is the Knowunity AI companion?
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
Where can I download the Knowunity app?
You can download the app from Google Play Store and Apple App Store.
Is Knowunity really free of charge?
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
Most popular content: Isotope
7Understanding Isotopes
Explore the concept of isotopes in chemistry, focusing on atomic number, mass number, and the differences in neutron count. This summary provides a clear definition and examples to enhance your understanding of isotopes and their significance in the study of elements.
Atomic Structure & Isotopes
Explore the fundamental concepts of atomic structure, including Dalton's atomic theory, the significance of atomic and mass numbers, and the role of subatomic particles. Understand isotopes and their characteristics, focusing on how atoms of the same element can differ in neutron count while maintaining identical proton numbers. This summary provides a clear overview for students studying chemistry.
WJEC AS-level Chemsitry Unit 1 Topic 3 Mass spectrometry
Summary of mass spectrometry and reading mass spectra’s. (NOTE: ignore the title saying Topic 1.4, topics were split differently in lessons to how the WJEC split them in text books)
Nat 5 chemistry unit 3
All information needed for chemistry nat 5
Understanding Isotopes & Ions
Explore the concepts of atomic number, mass number, isotopes, and ions in this detailed summary. Learn how protons, neutrons, and electrons define atomic structure and how variations in these subatomic particles lead to different isotopes and charged ions. Ideal for students studying chemistry fundamentals.
Understanding Isotopes in Chemistry
Explore the concept of isotopes, including their definitions, examples like Carbon-12 and Carbon-13, and how to calculate relative atomic mass using isotope abundance. This summary sheet is essential for AQA Chemistry students preparing for exams, highlighting key terms such as mass number and relative abundance.
Understanding Isotopes
Explore the fundamentals of isotopes, including definitions, calculations of relative atomic mass, and the relationship between protons, neutrons, and mass number. This summary covers key concepts such as relative abundance and atomic structure, essential for mastering Module 2 in Chemistry.
Most popular content in Chemistry
9GCSE AQA Chemistry Topic 1- The Atom
The Atom
chem paper 1
higher
A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
GCSE AQA Chemistry Topic 1
Quiz covering all of topic 1
Essential Chemistry Practicals
Explore key AQA GCSE Chemistry practicals, including flame tests, titration, and gas identification. This resource covers essential techniques for analyzing ions, making salts, and understanding reaction kinetics. Perfect for students preparing for exams and practical assessments.
chem paper 1
Unlock the secrets of chemical reactions with this comprehensive flashcard set designed to help you conquer complex concepts and ace your Chemistry exams.
AQA GCSE Chemistry 4.2 Bonding, structure and the Properties of Matter
4.2 Metals and Alloys
AQA GCSE Chemistry 4.4 Chemical changes
4.4 Reactions of acids with bases or carbonates
Fundamentals of Atomic Structure
Practice identifying subatomic particles, calculating neutrons, and defining isotopes and atomic/mass numbers.
Most popular content
9Sociology of Education Overview
Explore comprehensive A-Level Sociology notes on the education system, covering key theories, policies, and sociological perspectives. This resource includes insights on marketisation, gender roles, cultural deprivation, and educational inequalities, providing a thorough understanding of how education shapes social stratification and individual achievement. Ideal for exam preparation and in-depth study.
Sociology of Families: Comprehensive Revision
Dive into an extensive overview of family dynamics, perspectives, and patterns in sociology. This resource covers key concepts such as family diversity, gender roles, marriage, and the impact of social policies on family structures. Perfect for A-Level Sociology students preparing for Paper 2.
Criminology: Crime & Punishment Overview
Comprehensive mindmaps covering key concepts in the Crime and Punishment topic for WJEC Criminology Unit 4. This resource includes detailed insights into the Criminal Justice System, crime prevention strategies, sentencing models, and the roles of various agencies. Ideal for A-Level revision, ensuring you grasp essential theories and legislative processes to excel in your exams.
An Inspector Calls: Character Insights
Explore in-depth analysis and key quotes for characters in J.B. Priestley's 'An Inspector Calls'. This resource covers Gerald Croft, Inspector Goole, Sheila Birling, Mrs. Birling, Eric Birling, and Eva Smith, focusing on themes of class, gender roles, and social responsibility. Ideal for students aiming for Grade 8 and above.
WJEC Unit 4 Criminology
Criminology unit 4 detailed revision note
Criminology Theories Overview
Explore key criminology theories and their implications on crime and deviance. This comprehensive summary covers biological, psychological, and sociological perspectives, including labelling theory, right realism, and the impact of social campaigns on policy development. Ideal for A-Level criminology students seeking to understand the complexities of criminal behaviour and the factors influencing crime prevention strategies.
Romeo and Juliet: Key themes
Key Romeo and Juliet themes and analysed quotes
Macbeth: Guilt and Ambition
Explore the complex themes of guilt and ambition in Shakespeare's 'Macbeth'. This analysis covers key characters, including Macbeth and Lady Macbeth, their moral dilemmas, and the tragic consequences of their ambition. Ideal for students studying character motivations, thematic elements, and the psychological impact of power. Includes insights on the natural order, manipulation, and the descent into madness.
Cell Biology and Cell structure
cell structures
Can't find what you're looking for? Explore other subjects.
Students love us — and so will you.
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.
Understanding Atomic Structure and Isotopes for OCR A-Level Chemistry
Understanding atomic structure is absolutely crucial for mastering chemistry - it's the foundation that explains how elements behave and react with each other. This topic covers everything from the basic particles that make up atoms to sophisticated techniques scientists use... Show more

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Atomic Structure Basics
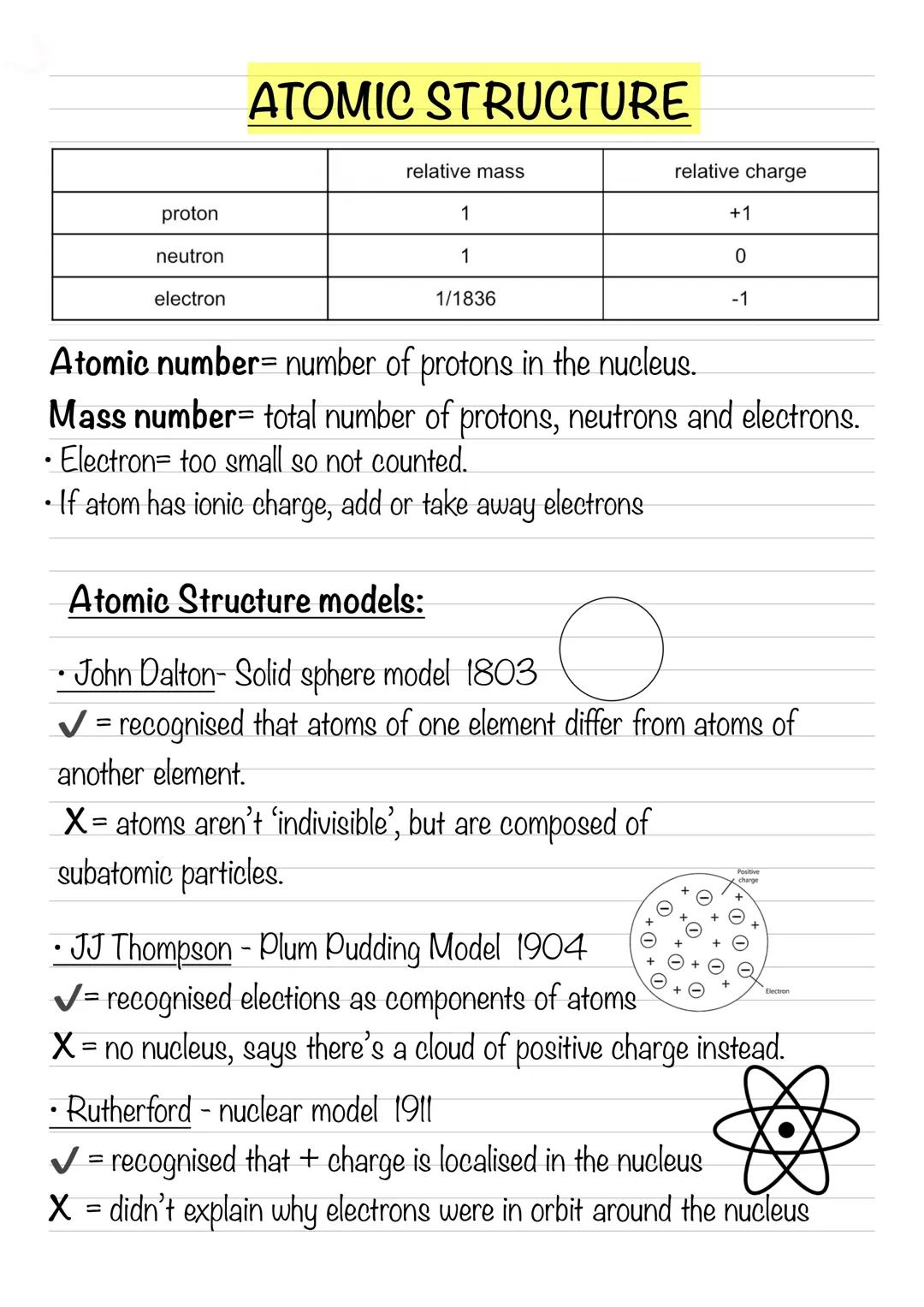
Every atom is made up of three fundamental particles that you need to know inside out. Protons have a relative mass of 1 and a charge of +1, neutrons also have a relative mass of 1 but are neutral (charge of 0), and electrons are tiny with a relative mass of 1/1836 and a charge of -1.
The atomic number tells you how many protons are in the nucleus - this never changes for a given element. The mass number is the total of protons and neutrons (electrons are too light to count). When dealing with ions, simply add or subtract electrons based on the charge.
Key Tip: Remember that protons define what element you're dealing with - change the number of protons, and you've got a completely different element!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Historical Models of the Atom
Scientists didn't always understand atomic structure - it took centuries of brilliant minds to figure it out! John Dalton (1803) imagined atoms as solid spheres, which was groundbreaking but missed the fact that atoms contain smaller particles.
JJ Thompson (1904) discovered electrons and proposed the plum pudding model - electrons embedded in a positive "pudding." Rutherford (1911) proved there's a dense nucleus through his famous gold foil experiment. Niels Bohr (1913) suggested electrons orbit in fixed paths, explaining why atoms don't collapse.
Finally, Schrödinger (1926) gave us the modern quantum model, where electrons exist in probability clouds called orbitals rather than fixed orbits. This model actually works for all atoms and is what we use today.
Remember: Each model built on the previous one's strengths whilst fixing its weaknesses - that's how science progresses!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Isotopes and Relative Mass
Isotopes are atoms of the same element with different numbers of neutrons - they're like identical twins with different weights! They have the same atomic number but different mass numbers, which means they behave almost identically in chemical reactions.
The extra neutrons don't affect chemical properties much, but they do change physical properties like melting points and densities. This is why hydrogen and deuterium (heavy hydrogen) have slightly different boiling points.
Relative atomic mass is the weighted average mass of all an element's isotopes compared to 1/12th of a carbon-12 atom. Relative isotopic mass refers to individual isotopes. These definitions might seem abstract, but they're essential for understanding why atomic masses aren't whole numbers.
Quick Check: If you see a non-whole atomic mass on the periodic table, you know that element has multiple isotopes!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Mass Spectrometry - The Basics
Mass spectrometry is like a sophisticated weighing scale that can identify and measure individual atoms and molecules. It tells you exactly what isotopes are present and in what percentages - incredibly useful for chemists!
The process starts by bombarding your sample with high-energy electrons, which knocks electrons off atoms to create positive ions. These ions get accelerated through an electric field, then separated based on their mass-to-charge ratio .
Since most ions have a +1 charge, the m/z ratio essentially just tells you the mass. A computer detects these separated ions and creates a mass spectrum - a graph showing what masses are present and how abundant they are.
Real-world Application: Mass spectrometry is used everywhere from detecting drugs in athletes to identifying unknown compounds in forensic investigations!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
How Mass Spectrometry Works
There are two main methods for separating ions in mass spectrometry. The magnetic deflection method uses a magnetic field to bend the path of ions - heavier ions get deflected less than lighter ones, allowing separation.
Time of flight spectrometry is more elegant - all ions get the same kinetic energy, so lighter ions move faster than heavier ones. By measuring how long it takes ions to travel a fixed distance, you can calculate their masses with incredible precision.
The final mass spectrum is straightforward to read: the x-axis shows relative isotopic mass, and the y-axis shows the percentage abundance of each isotope. The height of each peak tells you how common that particular isotope is.
Pro Tip: The tallest peak represents the most abundant isotope - this is often called the base peak!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Calculating Relative Atomic Mass
You'll definitely need to master these calculations for your exams! The formula is: Ar = Σ(relative isotopic mass × abundance) ÷ 100. This weighted average accounts for how common each isotope is.
For missing isotope masses, substitute 'x' for the unknown value, expand the brackets, multiply by 100, then solve the linear equation. It's just algebra with a chemistry twist!
When calculating percentage abundances, call one isotope 'x' and the other '100-x', then follow the same process. Your final percentages must always add up to 100% - if they don't, you've made an error somewhere.
Exam Success: Always double-check your percentages add to 100% and that your final atomic mass makes sense compared to the individual isotope masses!

Sign up to see the content. It's free!
- Access to all documents
- Improve your grades
- Join milions of students
Relative Molecular and Formula Mass
Calculating relative molecular mass and relative formula mass is refreshingly straightforward after all those isotope calculations! Simply add up the relative atomic masses of all atoms in the compound.
Use relative molecular mass for simple molecules (like H₂O or CO₂) and relative formula mass for compounds with giant structures like ionic compounds. The calculation method is identical - just different terminology.
Start by writing out all elements present, look up their relative atomic masses from the periodic table, multiply by how many of each atom you have, then add everything together. That's your final answer!
Time-Saver: Make a systematic list of elements and their quantities before calculating - it prevents silly mistakes and saves time in exams!
We thought you’d never ask...
What is the Knowunity AI companion?
Our AI Companion is a student-focused AI tool that offers more than just answers. Built on millions of Knowunity resources, it provides relevant information, personalised study plans, quizzes, and content directly in the chat, adapting to your individual learning journey.
Where can I download the Knowunity app?
You can download the app from Google Play Store and Apple App Store.
Is Knowunity really free of charge?
That's right! Enjoy free access to study content, connect with fellow students, and get instant help – all at your fingertips.
Most popular content: Isotope
7Understanding Isotopes
Explore the concept of isotopes in chemistry, focusing on atomic number, mass number, and the differences in neutron count. This summary provides a clear definition and examples to enhance your understanding of isotopes and their significance in the study of elements.
Atomic Structure & Isotopes
Explore the fundamental concepts of atomic structure, including Dalton's atomic theory, the significance of atomic and mass numbers, and the role of subatomic particles. Understand isotopes and their characteristics, focusing on how atoms of the same element can differ in neutron count while maintaining identical proton numbers. This summary provides a clear overview for students studying chemistry.
WJEC AS-level Chemsitry Unit 1 Topic 3 Mass spectrometry
Summary of mass spectrometry and reading mass spectra’s. (NOTE: ignore the title saying Topic 1.4, topics were split differently in lessons to how the WJEC split them in text books)
Nat 5 chemistry unit 3
All information needed for chemistry nat 5
Understanding Isotopes & Ions
Explore the concepts of atomic number, mass number, isotopes, and ions in this detailed summary. Learn how protons, neutrons, and electrons define atomic structure and how variations in these subatomic particles lead to different isotopes and charged ions. Ideal for students studying chemistry fundamentals.
Understanding Isotopes in Chemistry
Explore the concept of isotopes, including their definitions, examples like Carbon-12 and Carbon-13, and how to calculate relative atomic mass using isotope abundance. This summary sheet is essential for AQA Chemistry students preparing for exams, highlighting key terms such as mass number and relative abundance.
Understanding Isotopes
Explore the fundamentals of isotopes, including definitions, calculations of relative atomic mass, and the relationship between protons, neutrons, and mass number. This summary covers key concepts such as relative abundance and atomic structure, essential for mastering Module 2 in Chemistry.
Most popular content in Chemistry
9GCSE AQA Chemistry Topic 1- The Atom
The Atom
chem paper 1
higher
A-level OCR A Chemistry summary sheets
Everything from snaprevise for OCR chemistry a-level
GCSE AQA Chemistry Topic 1
Quiz covering all of topic 1
Essential Chemistry Practicals
Explore key AQA GCSE Chemistry practicals, including flame tests, titration, and gas identification. This resource covers essential techniques for analyzing ions, making salts, and understanding reaction kinetics. Perfect for students preparing for exams and practical assessments.
chem paper 1
Unlock the secrets of chemical reactions with this comprehensive flashcard set designed to help you conquer complex concepts and ace your Chemistry exams.
AQA GCSE Chemistry 4.2 Bonding, structure and the Properties of Matter
4.2 Metals and Alloys
AQA GCSE Chemistry 4.4 Chemical changes
4.4 Reactions of acids with bases or carbonates
Fundamentals of Atomic Structure
Practice identifying subatomic particles, calculating neutrons, and defining isotopes and atomic/mass numbers.
Most popular content
9Sociology of Education Overview
Explore comprehensive A-Level Sociology notes on the education system, covering key theories, policies, and sociological perspectives. This resource includes insights on marketisation, gender roles, cultural deprivation, and educational inequalities, providing a thorough understanding of how education shapes social stratification and individual achievement. Ideal for exam preparation and in-depth study.
Sociology of Families: Comprehensive Revision
Dive into an extensive overview of family dynamics, perspectives, and patterns in sociology. This resource covers key concepts such as family diversity, gender roles, marriage, and the impact of social policies on family structures. Perfect for A-Level Sociology students preparing for Paper 2.
Criminology: Crime & Punishment Overview
Comprehensive mindmaps covering key concepts in the Crime and Punishment topic for WJEC Criminology Unit 4. This resource includes detailed insights into the Criminal Justice System, crime prevention strategies, sentencing models, and the roles of various agencies. Ideal for A-Level revision, ensuring you grasp essential theories and legislative processes to excel in your exams.
An Inspector Calls: Character Insights
Explore in-depth analysis and key quotes for characters in J.B. Priestley's 'An Inspector Calls'. This resource covers Gerald Croft, Inspector Goole, Sheila Birling, Mrs. Birling, Eric Birling, and Eva Smith, focusing on themes of class, gender roles, and social responsibility. Ideal for students aiming for Grade 8 and above.
WJEC Unit 4 Criminology
Criminology unit 4 detailed revision note
Criminology Theories Overview
Explore key criminology theories and their implications on crime and deviance. This comprehensive summary covers biological, psychological, and sociological perspectives, including labelling theory, right realism, and the impact of social campaigns on policy development. Ideal for A-Level criminology students seeking to understand the complexities of criminal behaviour and the factors influencing crime prevention strategies.
Romeo and Juliet: Key themes
Key Romeo and Juliet themes and analysed quotes
Macbeth: Guilt and Ambition
Explore the complex themes of guilt and ambition in Shakespeare's 'Macbeth'. This analysis covers key characters, including Macbeth and Lady Macbeth, their moral dilemmas, and the tragic consequences of their ambition. Ideal for students studying character motivations, thematic elements, and the psychological impact of power. Includes insights on the natural order, manipulation, and the descent into madness.
Cell Biology and Cell structure
cell structures
Can't find what you're looking for? Explore other subjects.
Students love us — and so will you.
The app is very easy to use and well designed. I have found everything I was looking for so far and have been able to learn a lot from the presentations! I will definitely use the app for a class assignment! And of course it also helps a lot as an inspiration.
This app is really great. There are so many study notes and help [...]. My problem subject is French, for example, and the app has so many options for help. Thanks to this app, I have improved my French. I would recommend it to anyone.
Wow, I am really amazed. I just tried the app because I've seen it advertised many times and was absolutely stunned. This app is THE HELP you want for school and above all, it offers so many things, such as workouts and fact sheets, which have been VERY helpful to me personally.